Now is a really good time to focus on carbapenem-resistant bacteria. We have spent much of the past 3 years focusing on one particular virus. But now that the clinical issues linked to SARS-CoV-2 are waning for our hospital patients, the threat of carbapenem-resistance in Gram-negative bacteria comes to the fore. An excellent study with far-reaching consequences has been published from Italy. Carbapenem resistance is bad news if you have a BSI: patients with carbapenem-resistant BSI were roughly twice as likely to die as patients with carbepenem-susceptible BSI.
Continue readingKPC
Are our attempts to contain CPE going down the drain?
I posted recently on the potential risk of CPE contamination of sinks, drains, and hospital wastewater. The question in my mind then was whether contamination is a smoking gun or innocent bystander regarding CPE transmission? What we really need is an intervention to show that better management of sinks and drains results in reduce CPE transmission. And now, we have one! The findings suggest that attempts to control CPE will go down the drain if we don’t intervene to improvement the management of sinks and drains.
Looking back on a CPE plasmid attack in the Northwest of England
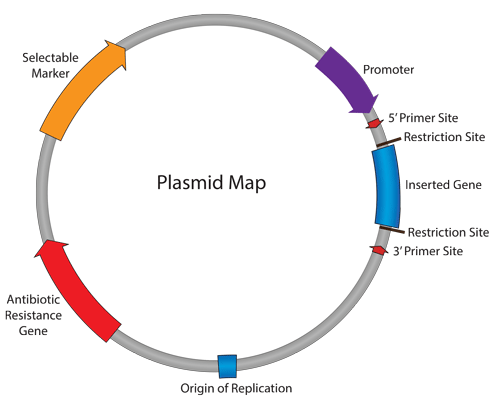
A genomic study of 44 isolates of CPE from various species identified between 2008 and 2010, mainly from the Northwest of England, has concluded that plasmids played a key role in the early dissemination of CPE.
Promiscuous plasmids: a rapid reflection from ECCMID 2017
I came to ECCMID 2017 with a very specific question: do we need to think beyond ‘same-bug-same-gene’ horizontal transmission from a practical IPC view point in order to address the threat of IPC? The answer, unfortuantely, is yes!
The carbapenemase is out there
A PNAS paper on the genomic diversity of carbapenemase producing bacteria in the US reports strong evidence of carbapenamase (an enzyme produced by bacteria that breaks down carabapenem antibiotics) activity but no sign of a carbapenemase gene. This provides a timely reminder that we are only really scratching the surface in our understanding of carbapenemases and how they work.
KPC Casanova carbapenemase
The risk of interspecies transmission of carbapenemase genes is a real concern. We can barely get our heads around many different types of carbapenemase in a whole host of Gram-negative bacteria (compare the relative simplicity of methicillin resistance in S. aureus: a single gene, in a single species). Throw in interspecies horizontal transmission of carbapenemases and things get really tricky! Do we implement different control strategies to try to interrupt the transmission of carbapenemases (in contrast to the organisms themselves)? Could you have a multispecies outbreak of a carbapenemase on your hands and not even realise it?
Is CRE lurking in nursing homes?
They say that things come in threes, so following hot on the heels of blogs about MRSA and other MDROs in nursing homes, I was struck by a recent outbreak report of CRE associated with nursing homes the Netherlands.
Following the admission of a patient from a Greek ICU, a nosocomial transmission of CRE (ST258 KPC K. pneumoniae) occurred. By the way, this occurred despite the hospital recognising the risk of CRE at the time of admission from the Greek ICU, perform an admission screening and implementing pre-emptive contact precautions. Then the index patient was transferred to a nursing home, where subsequent transmission occurred to four other patients.
CRE “trafficking” plasmids through hospital surfaces
A team from the NIH Clinical Center in the US present a fascinating study, exploring the transmission of carbapenemase-encoding plasmids in unprecedented detail. The intro does a good job of introducing the ‘triple threat’ from CRE: pan-drug resistance, sharply increasing prevalence, and the potential for the horizontal transfer of carbapenemase genes between Enterobacteriaceae species. They introduce the idea of “plasmid trafficking”, which evokes images of shady bacteria dealing in antibiotic resistance genes (a la the infamous cartoon below):
NIH is a hospital that takes CRE seriously, after being stung by an outbreak in 2011. A quick look at who they screen for CRE illustrates just how seriously they take the threat:
- ICU / high-risk patients screened twice weekly.
- All patients screened monthly.
- Admissions from other hospitals screened for CRE…twice (and given pre-emptive contact precautions until negative cultures are confirmed, for good measure).
They also performed some environmental sampling and recovered several CRE from the hospital environment. This will surprise some, but Enterobacteriaceae do have the potential to survive on surfaces for longer than you may expect.
Surveillance cultures identified 10 patients with KPC-producing Enterobacteriaceae and environmental surveillance identified 6 KPC-producing Enterobacteriaceae. They combined these with several historic isolates from the 2011 outbreak, and a couple of imported isolates to give a sample size of 20 isolates. They wanted to dig deeper into these isolates to explore whether or not they shared any plasmids. And here’s where it gets rather complicated. Conventional whole genome sequencing produces many short reads (100-500 bp) but these cannot distinguish between plasmids and chromosome-encoded genes. Therefore, the authors used a technique called single-molecule, real-time (SMRT) to generate longer reads (around 1000 bp) that make it possible to distinguish between plasmids and chromosome-encoded genes. [I know that I’ve over-simplified this clever genomics massively – but I’ll quickly get out of my depth otherwise!]
The report presents a picture of rare patient-to-patient nosocomial transmission (only 1 of 10 patients were thought to be in-hospital acquisitions), continual importation of diverse CRE, and a complex network of even more diverse plasmids. To illustrate the diversity, one strain of CRE contained no fewer than three distinct KPC-encoding plasmids!
The authors find some evidence of environmental spread of carbapenemase-encoding plasmids, with the carbapenemase-encoding plasmid from a patient matching plasmids recovered from different species of Enterobacteriaceae found in the patient’s environment. What the authors did not demonstrate is transmission of carbapenemase-encoding plasmids from the environment to patients – but I wouldn’t want to be admitted to a room with CRE lurking in the hospital environment!
There’s quite a bit of science around the horizontal transmission of plasmids within biofilms. Combine this with the recent finding of biofilms on dry hospital surfaces, and you have a concerning new angle on how CRE may be transmitted in hospitals.
Image credit. Nick Kim, with permission.
Article citation: Conlan S, Thomas PJ, Deming C et al. Single-molecule sequencing to track plasmid diversity of hospital-associated carbapenemase-producing Enterobacteriaceae. Sci Transl Med 2014; 6: 254ra126.
Dissecting the epidemiology of resistant Enterobacteriaceae and non-fermenters
It was a great privilege to speak at the HIS / IPS Spring Meeting today. You can download my slides here. The meeting was entitled: “What’s that coming over the hill? Rising to the challenge of multi-resistant Gram-negative rods”. This, I think, is an (oblique) reference to the signature hit of a Welsh band ‘The Automatic’: “What’s that coming over the hill? Is it a monster?”. So, are multi-resistant Gram-negative rods monsters lurking underneath the bed? Dr Tom Frieden, CDC Director, has described CRE as “nightmare bacteria” and Dr Sally Davies, CMO, has painted a bleak post-antibiotic era picture in reference to the emergence of these bacteria. So, is it a monster? Yes, I think it probably is. But all monsters are not created equal…
My exploration of the differences in the epidemiology of resistant Enterobacteriaceae and non-fermenters (mainly A. baumannii) was designed to prompt anybody tempted to conflate these two related problems to think twice. Resistant Enterobacteriaceae and non-fermenters do share the same response to the Gram-stain and can be resistant to key antibiotics occasionally through shared mechanisms (principally the carbapenemases). But that’s about it. Otherwise they’re like chalk and cheese. (A. baumannii = chalk, which turns to dust; Enterobacteriaeae = a good cheese, which ultimately ends up in the gut.) (Table).
Table: Comparing the epidemiology of resistant Enterobacteriaceae and non-fermenters.
| Enterobacteriaceae (K. pneumoniae) | Non-fermenters (A. baumannii) | |
| At-risk population | Primarily acute pts | ICU, burns |
| Risk factors | Travel | Trauma, ICU stay |
| Epidemic potential | High | Low |
| Clinical | UTI | VAP |
| Mortality | Stark increase (CPE) | Minimal increase |
| Prevalence | Emerging (rapidly) | Patchy but stable |
| Sites of colonisation | GI tract | Skin, resp & GI |
| Colonization duration | Months to >1 year | Days to weeks |
| Transmission routes | Hands ++, Env +/- | Hands +, Env ++ |
| Resistance | Mainly acquired | Intrinsic & acquired |
| Common clones | KPC-producing ST258 | Intl clones I-III |
Probably the most important difference between the Enterobacteriaceae and the non-fermenters is their at-risk populations. A. baumannii is restricted mainly to high-risk patients in intensive care units. This is not so for the resistant Enterobacteriaceae, which have the potential to cause infection and colonization in a wide group of hospitalized patients and, perish the thought, the community. Related to this is their epidemic potential: resistant Enterobacteriaceae, including CRE, have already demonstrated the capacity to spread rapidly and broadly in hospital and to a lesser extent community populations. Rates of antibiotic resistance in A. baumannii can be high, but it does not possess the tools to spread outside of high-risk hospitalized patients.
Other differences between these two groups of resistant Gram-negative bacteria include the types of infection they cause and associated attributable mortality, their prevalence, their sites and duration of colonization, their transmission routes, their resistance mechanisms and their population structure and clonal dissemination patterns (Table).
One of the many acronyms in current circulation to describe resistant Gram-negatives is CRO (carbapenem-resistant organisms), which is used as a catch-all term to encompass both Enterobacteriaceae and non-fermenters. Given the fundamental differences in epidemiology, I don’t think it’s very helpful. After all, MRSA is a ‘CRO’ but we wouldn’t dream of lumping it together with K. pneumoniae or A. baumannii! So, we should expunge ’CRO’ from our collective vernacular and stick to CRE and CRAB.
I accept that there are limitations with my presentation. You could (and probably should) further dissect the epidemiology of K. pneumoniae vs. E. coli, A. baumannii vs. P. aeruginosa, and ESBL vs. carbapenemase producers. I suspect we will eventually have data to demonstrate different clinical manifestations associated with the various common carbapenemase genes.
That said, I think a separation of the resistant Enterobacteriaceae and non-fermenters is a useful distinction in terms of at-risk populations, epidemic potential, and effective control measures.
Finally, my preparation for the talk raised several challenging questions:
- Which interventions work?
- Are they different for Enterobacteriaceae and non-fermenters? (Probably, given their epidemiology.)
- Has our focus on CPE taken our eye off CPNF, which are the ‘clear and present danger’ for many of us?
- What is the prevalence of CPE in the UK?
- How much do we believe a single negative screen?
- Do we need rapid molecular diagnostics?
- What is the duration of colonisation?
- Are there decolonisation strategies other than “selective” decontamination using antibiotics?
Image credit: ‘Chalk and Cheese’ by Jackson Boyle.
Dissecting the CRE epidemic in Italy
 Carbapenem-resistant Enterobacteriaceae (CRE) present unique challenges to infection prevention and control. Firstly, unlike MRSA and C. difficile, CRE can be caused by multiple genetic determinants (typically KPC, VIM, NDM and OXA-48 types) in multiple species. The combination of resistance determinants and species may have distinct characteristics with transmission and control implications. Further, there is a larger pool of resistance determinants for horizontal transfer. Secondly, CRE colonize the gastrointestinal tract, so deconlonization therapy is likely to be limited to suppressing the amount of CRE in the gut; elimination of the carrier state, which has been a mainstay of prevention and control interventions for MRSA, seems unlikely. Thirdly, pan-drug resistant CRE has already been reported and the pipeline for new agents is virtually empty, meaning that effective therapeutic options will be increasingly limited.
Carbapenem-resistant Enterobacteriaceae (CRE) present unique challenges to infection prevention and control. Firstly, unlike MRSA and C. difficile, CRE can be caused by multiple genetic determinants (typically KPC, VIM, NDM and OXA-48 types) in multiple species. The combination of resistance determinants and species may have distinct characteristics with transmission and control implications. Further, there is a larger pool of resistance determinants for horizontal transfer. Secondly, CRE colonize the gastrointestinal tract, so deconlonization therapy is likely to be limited to suppressing the amount of CRE in the gut; elimination of the carrier state, which has been a mainstay of prevention and control interventions for MRSA, seems unlikely. Thirdly, pan-drug resistant CRE has already been reported and the pipeline for new agents is virtually empty, meaning that effective therapeutic options will be increasingly limited.
Data from EARS-Net suggests that the prevalence of CRE among bloodstream infections is low in most parts of Europe, with a gradual year-on-year increase. In Greece though, rates are exceptionally high, with the proportion of K. pneumoniae invasive isolates resistant to carbapenems increasing from 27.8% in 2005 to 68.2% in 2011. Also, rates in Cyprus are on the rise with 0% reported in 2006 up to to 15.7% in 2011. In the UK, rates of carbapenem resistance amongst K. pneumoniae have remained consistently <1% for the same period. Disturbingly, there has been a dramatic increase in the prevalence of carbapenem-resistant K. pneumoniae in the last few years in Italy, from 1% in 2009 to 15% in 2010 to 27% in 2011.
 [Chart: Changes in proportion of carbapenem resistance in K. pneumoniae invasive isolates. Data from EARS-Net.]
[Chart: Changes in proportion of carbapenem resistance in K. pneumoniae invasive isolates. Data from EARS-Net.]
A recent Eurosurveillance article reports a national survey of carbapenem resistance in Italy. 25 laboratories across the country participated and analyzed all consecutive, non-duplicate Enterobacteriaceae clinical isolates for six weeks in mid 2011. A total of 7,154 isolates were collected from inpatients and 6,595 isolates from outpatients. The highlight findings are:
- 3.5% of inpatient isolates and 0.3% of outpatient isolates carbapenem resistant.
- Carbapenem-resistant K. pneumoniae (CR-KP) the most problematic CRE, with 11.9% of K. pneumoniae isolates CR.
- Substantial geographical variation in resistance rate, ranging from 0 to 33% for CR-KP.
- KPC accounted for 90% of CRE enzymes; one CR-KPC clone predominated (CC-258).
- Resistance to other agents was common amongst KPC-producing K. pneumoniae; 22% were resistant/non-susceptible to colistin, 21% to tigecycline and 16% to gentamicin; 1.5% were non-susceptible to all three.
This study raises several challenging questions. What do you do with a CR-KP isolate causing an infection that is also resistant to colistin, gentamicin and tigecycline? This seems to be true pan-resistance, with supportive care the only option.
Why is KP the outstanding CRE, specifically the CC-258 clone? What does it have that the other CRE lack? K. pneumoniae seems to survive better on surfaces that other Enterobacteriaceae, and has been associated with more hospital outbreaks than other Enterobacteriaceae historically. However, further research is required to answer this question.
Can the worrying trend of CRE in Italy be reversed? An aggressive, national intervention was successful in Israel, and there are some local success stories in Italy. However, brining the situation under control in Italy will require an aggressive, national programme that must be implemented immediately. Otherwise, CR-KP will quickly become endemic and probably impossible to bring under control.
The authors should be complimented for performing a timely study, but I do wonder whether the situation is considerably worse now, 12 months later, given the shape of the national epi curve.
Citation: Giani T, Pini B, Arena F, Conte V, Bracco S, Migliavacca R, the AMCLI-CRE Survey Participants, Pantosti A, Pagani L, Luzzaro F, Rossolini GM. Epidemic diffusion of KPC carbapenemase-producing Klebsiella pneumoniae in Italy: results of the first countrywide survey, 15 May to 30 June 2011. Euro Surveill 2013;18(22):pii=20489.
Image permission: Original image obtained from http://www.freestock.ca.