Ever found yourself wondering how often colonization precedes infection with MDR-GNB and VRE? A new systematic review and meta-regression in Lancet ID gives us a pretty solid answer: about 14% of MDR-GNB and 8% for VRE. This information is helpful for us to qualify the significance of acquiring these organisms in healthcare settings.
Continue readingESBL
Ethnicity and infection: avoiding spurious associations
I’ve been meaning to blog on this editorial about ethnicity as a risk factor (or not!) for infection by Prof Leibovici in Clinical Microbiology and Infection for a while. The basic story is that “ethnicity” often falls out as associated with infection-related variables (e.g. colonisation or infection with resistant bacteria), but the editorial poses an important question: it is actually ethnicity, or something co-correlated with ethnicity (e.g. socio-economic status) that is the causal risk factor?
Single rooms for ESBLs anyone?
A comprehensive and impressive cluster randomised crossover study published in Lancet ID examines whether it makes sense to use single rooms (as compared with multi-bed bays) to apply contact precautions for patients known to be carrying ESBL-Enterobacteriaceae. I need to be careful what I say, because fellow bloggers Marc and Andreas are co-authors. However, the gist seems to be: don’t bother with single rooms for ESBL-E carriers – but many hospitals don’t have capacity to do that anyway, so this may not be a practice-changing finding in many parts of the world!
Contact Precautions for Endemic MRSA and VRE
 by Andreas Voss and Eli Perencevich,
by Andreas Voss and Eli Perencevich,
intentionally posted on “Reflections” and “Controversies” at the same time as a reaction to the JAMA Viewpoint by Morgan, Wenzel & Bearman
During the recent ICPIC 2017 and a pre-meeting think tank, the sense and non-sense of RCTs looking at various infection control measures was a major point of discussion during many sessions. Data from well-designed quasi-experimental studies, epidemiological evidence, and logic seems to vanish, whenever a new RCT is published, even if the results are not applicable to situations that are non-endemic, have higher or lower compliance with the preventive measures in question, or whether the intended measures were actually applied within the intended patient group. Some studies seem to assume that the transmission during the first days of admission are of no consequence. Others assume that given endemicity and a high patient load, the intended measures such as single-room isolation can’t be applied, even if a patient was randomized to receive those measures.
Staff screening for MDROs: closing Pandora’s Box
A brave study from the Palmore/Frank group at NIH has opened the Pandora’s Box that is screening staff for MDROs, and, I’m delighted to say, firmly closed it with their findings! Only 3% of staff carried ESBLs, one carried a CPE, and none carried VRE, and this despite extensive contact with MDRO patients for many of the staff sampled!
Can we halve Gram-negative BSIs by 50% by 2021? The crowd say “No”
Most of those casting their vote supported Martin’s (somewhat pessimistic) view that we can’t halve Gram-negative BSI by 2021 (see the figure, below). Let me first give you my own, unspoiled opinion (written before the results of this survey were known). I was intending to vote for option 3 (the English can’t, the Dutch might) but I am not even sure of that; actually, I believe that neither the English nor the Dutch can.
Antibiotic Awareness: background and links
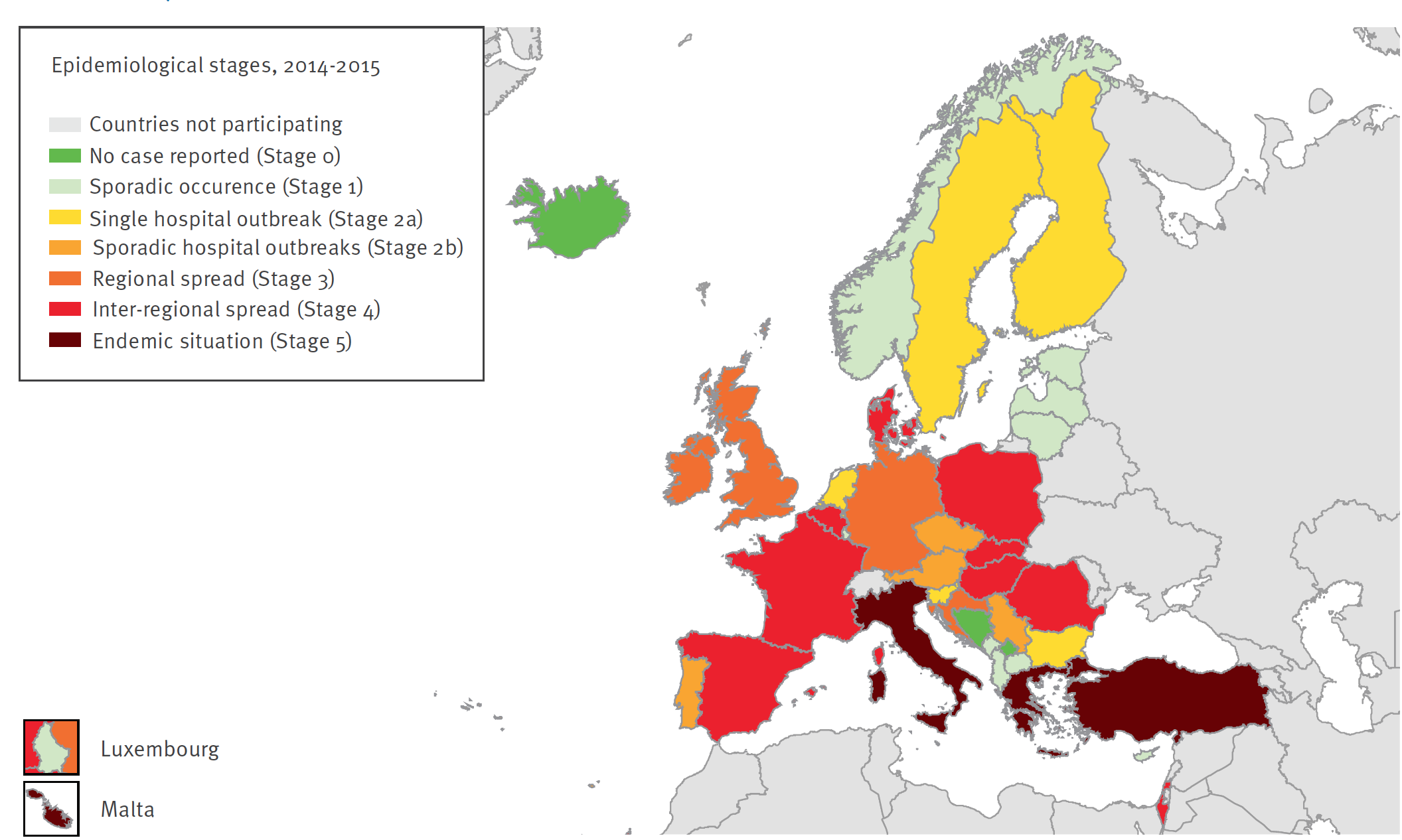
European Survey of Carbapenemase-Producing Enterobacteriaceae (EuSCAPE) project
The EuSCAPE project aimed to improve understanding of the occurrence and spread of CPE. LINK
About European Antibiotic Awareness Day
European Antibiotic Awareness Day is a European health initiative coordinated by ECDC which aims to provide a platform and support for national campaigns on the prudent use of antibiotics. Each year across Europe, the European Antibiotic Awareness Day is marked by national campaigns on the prudent use of antibiotics during the week of 18 November. Prudent use means only using antibiotics when they are needed, with the correct dose, dosage intervals and duration of the course. Follow the European Antibiotic Awareness Day: #EAAD. http://antibiotic.ecdc.europa.eu
About World Antibiotic Awareness Week
The World Health Organization is leading a global campaign for the first World Antibiotic Awareness Week with the slogan “Antibiotics: Handle with Care”. The campaign calls on individuals, governments and health and agriculture professionals to take action to address this urgent health problem. The first World Antibiotic Awareness Week will take place on 16-22 November. Follow the World Antibiotic Awareness Week: #AntibioticResistance. www.who.int/drugresistance.
Continue reading
More of a bad thing: ESBL-E
In a short, but important Dutch study, the added value of selective pre-enrichment for the detection of ESBL-producing enterobacteriacea (ESBL-E) was evaluated. The authors used their yearly prevalence study to shed more light onto the question if pre-enrichment (using a broth) might be equally improving the performance of ESBL-E detection, as it does with MRSA. While the literature on the topic might be controversial, this straightforward, well-performed study showed that direct culture failed to identify 25.9% (7/27) ESBL-E rectal carriers, which corresponds to 1.2% (7/562) of the screened population. While the overall rate of ESBL-E rectal carriage is not very high (4.8%) this study still demonstrates the importance of improving our methods to detect multi-drug resistant pathogens.
Do you know your CRE from your CRAB?
I gave a talk today at a meeting on combating carbapenem-resistant organisms. My angle was to clearly differentiate the epidemiology of the Enterobacteriaceae (i.e. CRE) from the non-fermenters (most importantly carbapenem-resistant A. baumannii – CRAB), and you can download my slides here.
I’ve blogged before about how confusing the terminology surrounding multidrug-resistant Gram-negative rods has become. Non-expert healthcare workers have little chance in distinguishing CRE from CPE from CRO from CPO. So we need to help them by developing some clear terminology, given the gulf in epidemiology between CRE and CRAB (see below).
CRE and CRAB are like apples and pears: they share some basic microbiology but that’s about where the comparison ends!
So, I think we should talk in terms of CRE (and CPE for confirmed carbapenemase carriers), and CRNF (or CRAB for A. baumannii and CRPA for P. aeruginosa). I don’t think that CRO is a useful term – in fact, I find it rather confusing. Carbapenem resistance in Enterobacteriaceae (CRE) and A. baumannii (CRAB) are both emerging problems, but they are not the same problem.
Not all resistant Gram-negative bacteria are created equal: Enterobacteriaceae vs. non-fermenters
Apples and oranges. They’re both more or less spherical and classified as fruits, and that’s about whether the similarity ends. It’s the same for antibiotic-resistant Enterobacteriaceae (e.g. Klebsiella pneumoniae) and non-fermenters (e.g. Acinetobacter baumannii): they both share the same basic shape (more or less) and classification (Gram-negative), and that’s about where the similarity ends (see the Table below):
Table: Comparing the epidemiology of resistant Enterobacteriaceae and non-fermenters.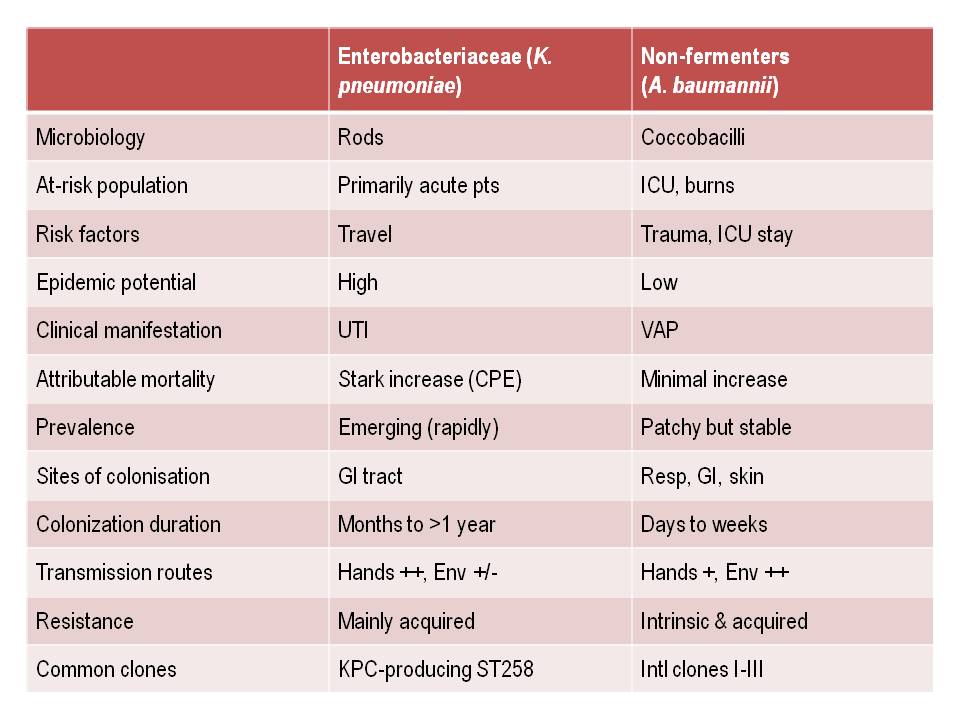
I gave a webinar yesterday as part of a three part series on resistant Gram-negatives. You can download the slides here, and access the recording here (although you’ll have to register to do so). I am increasingly hearing people talking about ‘carbapenem-resistant organisms’ (CRO), used as a catch-all term to encompass both the Enterobacteriaceae and the non-fermenters. As you can see from the comparison table able, this doesn’t make a lot of sense given the key differences in their epidemiology. Indeed, MRSA is a CRO, so why don’t we lump that together with the Enterobacteriaceae and non-fermenters? Carbapenem-resistant Enterobacteriaceae and carbapenem-resistant non-fermenters are both emerging problems, but they are not the same problem.
I asked a few questions of the audience, which I’ve summarised below:
Figures: Questions asked of around 150 webinar participants, mainly from the USA.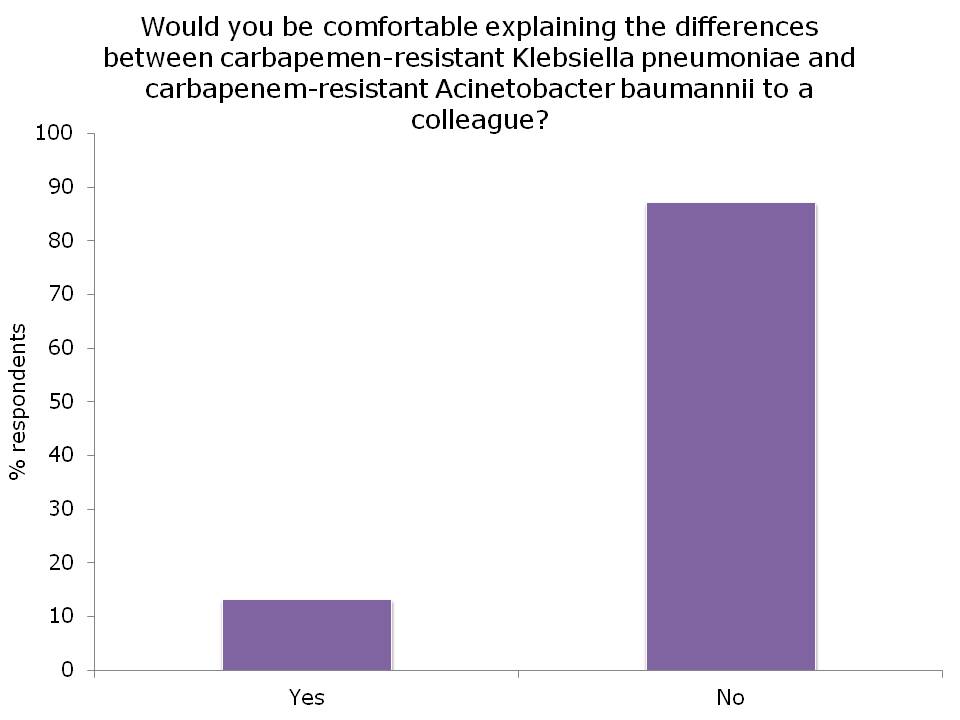

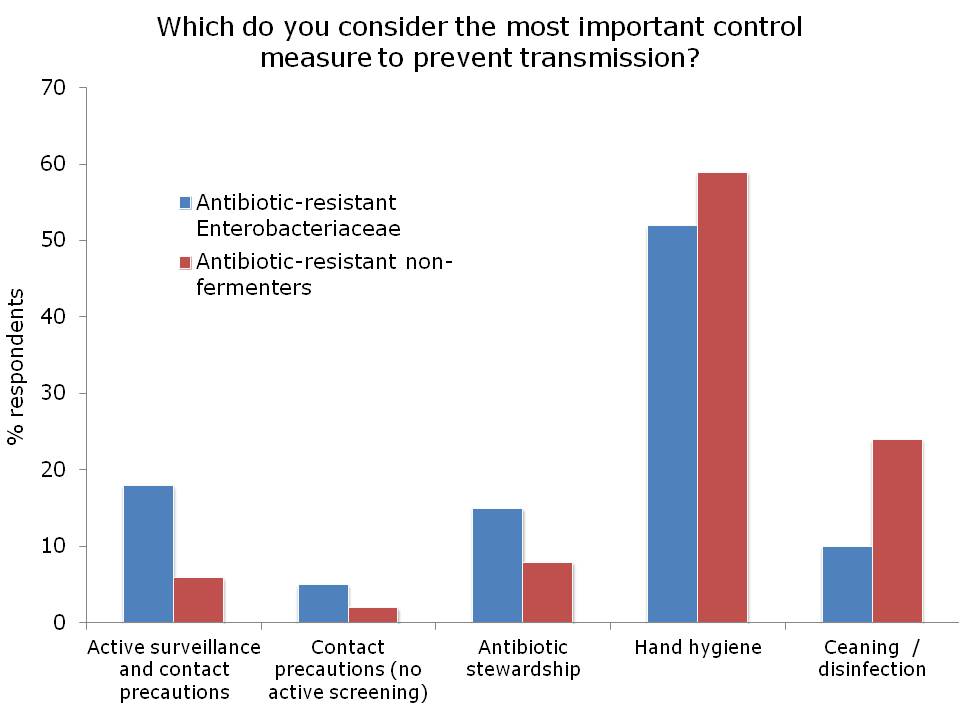
I was not surprised that so few people felt comfortable explaining the difference between the Enterobacteriaceae and non-fermenters – and this rather justified the whole thrust of the webinar! I was a little surprised that the ‘prevalence’ of the two groups of resistant bacteria were so similar; I was expecting the Enterobacteriaceae to be more common (although I admit this wasn’t a brilliantly worded question). In terms of control interventions, it’s true that we still don’t really know what works to control resistant Gram-negative bacteria. But it does seem likely that the control interventions will be different for Enterobacteriaceae and non-fermenters, and this did come across in the responses. Hand hygiene was selected by most people (which makes sense), with screening & isolation, and stewardship more commonly selected for Enterobacteriaceae, and cleaning / disinfection for the non-fermenters.
Q&A
Following the webinar, the audience asked a few interesting questions:
- Can you get chlorhexidine resistant organisms? A number of studies have hinted that reduced susceptibility to chlorhexidine may be an emerging problem, (for example Batra, Otter, Lee and Suwantarat). But increases in bacterial MICs (for Gram-positive bacteria at least) appear to be a long way below the applied concentration. However, it’s worth noting that the measured CHG skin concentration in one study (15-312 mg/L before the daily bath and 78-1250 mg/L after the daily bath) was much lower than the applied CHG concentration (10,000 mg/L). This is around the CHG MIC for some Gram-negatives and potentially brings the subtly reduced susceptibility to CHG reported in MRSA into play. On balance though, the rationale and data on reduced susceptibility are cautionary but not enough to recommend against universal use in the ICU given the clinical upside.
- Do you think we should be doing universal chlorhexidine bathing? On our ICU in London, we have been using universal chlorhexidine decolonization for a decade combined with targeted screening and isolation, and have seen a dramatic reduction in the spread of MRSA. So yes, I think we should be doing universal chlorhexidine bathing, but the need to monitor carefully for the emergence of clinically-relevant reduced susceptibility.
- Can we discontinue contact precautions for CRE? The short answer is no. Quite a few studies have found that gut colonization with CRE typically lasts for at least 6 months to >1 year. And those that become spontaneously ‘decolonised’ sometimes revert to colonized, suggesting that they weren’t really decolonized at all – it’s just that their load of CRE at the time of sampling had fallen below the limit of detection. So I favour a “once positive, always positive” approach to CRE colonization.
- Which disinfectant would you recommend for resistant Gram–negatives? It does seem that the non-fermenters (and in particular A. baumannii) are “more environmental” than the Enterobacteriaceae. However, the Enterobacteriaceae (including CRE – especially K. pneumoniae) can survive on dry surfaces for extended periods. Therefore, I think enhanced disinfection – especially at the time of patient discharge – is prudent for both groups. Consider using bleach or hydrogen peroxide-based liquid disinfectants, and terminal disinfection may be a job for automated room disinfection systems, such as hydrogen peroxide vapour.
- Should we use objective tools to monitor cleaning? Effective tools are available to objectively monitor cleaning (e.g. ATP and fluorescent dyes), and these have been shown to improve surface hygiene. Therefore, we should all now be using these tools to performance manage our cleaning processes.
Image credit: ‘Apples and oranges’.



