I have posted before about the “postcode lottery” and it’s influence on rates of MRSA, affecting deprived postcodes (or zipcodes!) more than others. But this issue is wider than regional influence. On an international scale, your changes of a bad outcome from infectious disease varies, sometimes wildly, by where you happen to be in the world. A new article in CID on clinical outcomes from carbapenem-resistant Acinetobacter baumannii infections bears this out.
Continue readingAcinetobacter
The infinite trio from South Africa
Last week I had the pleasure of attending the 8th FIDSSA Congress in Johannesburg (Federation of Infectious Diseases Societies of Southern Africa). I was invited to talk on infection control in the Netherlands, SDD and empiric antibiotic strategies in ICU. I never felt more distance between my habitat and that of my hosts. It surpassed the 3732 miles in the air. I learned a lot; from how it is to go into military conflict areas to identify Ebola cases, fighting a cholera outbreak after a tropical cyclone in Mozambique to the infinite trio, which stands for carbapenem resistant Klebsiella, Pseudomonas and Acinetobacter. Continue reading
The antibiotic resistance crisis resolved by bateriophages (part 5)
Yesterday we had another episode on the miracles of bacteriophage therapy on Dutch television. In the show I asked our minister of Health to modify the Dutch law, in order to make scientific evaluation of this approach in patients possible. Yet, the reasons why we need this change were not broadcasted. As several patients explained how they had been treated across Europe, this must have bee confusing, people not understanding why I – the knucklehead – failed to do studies. Let me explain. Continue reading
AMR deaths in Europe & America
Just before Christmas a follow-up on that what bothers us most: patients dying because of antibiotic resistance. I previously tried, see here, to disentangle from the ECDC study (33.000 deaths per year in Europe) how they got to 206 AMR casualties in the Netherlands and ended with a recommendation to not “focus too much on the absolute numbers as they may not be very precise.” With Valentijn Schweitzer I spent some more time in the 200 pages supplement, only to find out – in the end – that the Americans do these kind of studies much better. Continue reading
The reality of AMR in Greece
This week I attend the general assembly of COMBACTE, this year in Athens. COMBACTE stands for COMBatting AntibiotiC resistance in Europe (www.combacte.com) and is part of the New Drugs for Bad Bugs (ND4BB) program of the Innovative Medicines Initiative. Our local host is professor George Daikos, who opened the meeting with an overview of the epidemiology of antibiotic resistance in his country. Continue reading
Attacking the fecal veneer*
What an excellent start of 2017. A great study from the USA today in Lancet: In a pragmatic cluster-randomized crossover study they tested 4 patient room cleaning strategies on the effectiveness to reduce acquisition with relevant bacteria for the incoming patients. The conclusion states that “enhanced terminal room disinfection decreases the risk of pathogen acquisition.” Yet, this paper is so “data-dense” that you must read the methods (and supplements) to get the picture. In one shot: Not for C. diff, may be for MRSA and yes for VRE. Continue reading
Do you know your CRE from your CRAB?
I gave a talk today at a meeting on combating carbapenem-resistant organisms. My angle was to clearly differentiate the epidemiology of the Enterobacteriaceae (i.e. CRE) from the non-fermenters (most importantly carbapenem-resistant A. baumannii – CRAB), and you can download my slides here.
I’ve blogged before about how confusing the terminology surrounding multidrug-resistant Gram-negative rods has become. Non-expert healthcare workers have little chance in distinguishing CRE from CPE from CRO from CPO. So we need to help them by developing some clear terminology, given the gulf in epidemiology between CRE and CRAB (see below).
CRE and CRAB are like apples and pears: they share some basic microbiology but that’s about where the comparison ends!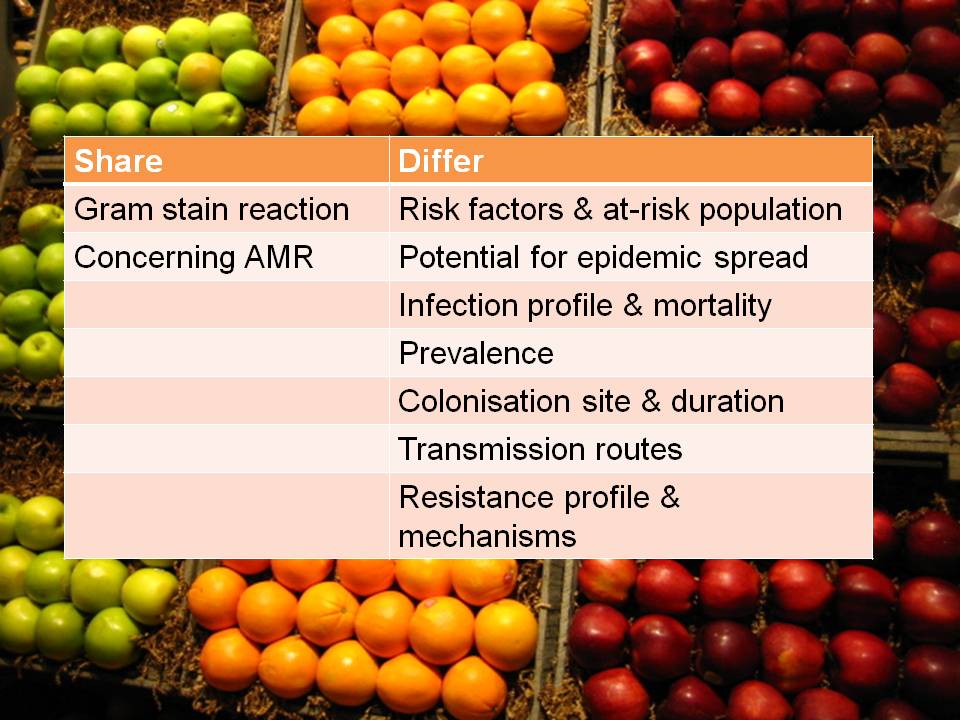
So, I think we should talk in terms of CRE (and CPE for confirmed carbapenemase carriers), and CRNF (or CRAB for A. baumannii and CRPA for P. aeruginosa). I don’t think that CRO is a useful term – in fact, I find it rather confusing. Carbapenem resistance in Enterobacteriaceae (CRE) and A. baumannii (CRAB) are both emerging problems, but they are not the same problem.
Does chlorhexidine bathing work for Gram-negative bacteria?
 The idea of “source control” – using chlorhexidine to reduce the amount of bacteria on a patient’s skin – makes a lot of sense. There’s mounting evidence that chlorhexidine daily bathing works for Gram-positive pathogens, especially in the ICU.1 For example, one of the first thorough studies of chlorhexidine gluconate (CHG) daily bathing showed that the amount of VRE on the skin, in the environment and transmitted to others were all reduced by implementing CHG daily bathing.2 A number of more recent high-quality studies have provided evidence that CHG daily bathing in the ICU setting helps to prevent the transmission of Gram-positive bacteria (see Table below – although note that studies have not been universally positive for CHG).
The idea of “source control” – using chlorhexidine to reduce the amount of bacteria on a patient’s skin – makes a lot of sense. There’s mounting evidence that chlorhexidine daily bathing works for Gram-positive pathogens, especially in the ICU.1 For example, one of the first thorough studies of chlorhexidine gluconate (CHG) daily bathing showed that the amount of VRE on the skin, in the environment and transmitted to others were all reduced by implementing CHG daily bathing.2 A number of more recent high-quality studies have provided evidence that CHG daily bathing in the ICU setting helps to prevent the transmission of Gram-positive bacteria (see Table below – although note that studies have not been universally positive for CHG).
Table: Studies evaluating the impact of chlorhexidine daily bathing (with or without other interventions) including data on Gram-negative bacteria.
| Study | Setting | Design | Intervention | Results |
| Noto 2015 3 | ICU | Cluster RCT | Daily CHG | No significant reduction in HCAI (composite measure including CLABSI, CAUTI, VAP and CDI) |
| Derde 2014 4 | ICU | Time series analysis | Daily CHG plus hand hygiene | Reduction in all MDROs and MRSA (but not VRE or ESBLs) |
| Seyman 2014 5 | ICU | Before-after | Weekly CHG ‘douche’ | Reduction in BSI but not CLABSI; slight reduction in Gram-negative BSI (n too small for statistical analysis) |
| Hayden 2014 6 | LTAC | Before-after | Bundle (including daily CHG) | Acquisition of CRE fell from 4 to 2 per 100 patient weeks |
| Martínez-Reséndez 2014 7 | ICU | Before-after | Daily CHG plus hand hygiene | Reductions in all infections, and in A. baumannii VAP rate |
| Apisarnthanarak 2014 8 | ICU | Before-after | Bundle (including daily CHG) | Reductions in a. baumannii infection and colonization |
| Climo 2013 9 | ICU | Cluster RCT | Daily CHG | Reductions in MRSA / VRE acquisition and all BSI; BSI mainly CoNS (no significant reduction in Gram-negative BSI or CLABSI) |
| Milstone 2013 10 | Paed ICU | Cluster RCT | Daily CHG | BSI reduced; mainly CoNS (no significant reduction in Gram-negative BSI or CLABSI) |
| Munoz-Price 2010 11 | LTAC | Before-after | Bundle (including daily CHG) | CRE carriage prevalence fell from 21% to 0% |
| Evans 2010 12 | ICU | Before-after | Daily CHG | Rate of CLABSI reduced; A. baumannii colonisation reduced but not significantly |
| Popovich 2009 13 | ICU | Before-after | Daily CHG | Rate of CLABSI reduced; A. baumannii infection rate reduced but not significantly |
| Bleasdale 2007 14 | ICU | Cross-over | Daily CHG | Number of Gram-negative BSI in each arm too small for analysis |
| Gould 2007 15 | ICU | Before-after | Daily CHG + mupirocin | Number of Gram-negatives too small for analysis |
| Camus 2005 16 | ICU | RCT | Daily CHG + mupirocin | Number of Gram-negative acquisitions similar in intervention vs. control groups |
The question of whether CHG is effective for the prevention and control of Gram-negative bacteria is rather more complicated. The main issue is that Gram-negative bacteria are less susceptible to CHG than Gram-positive bacteria.17 In theory, this shouldn’t be a problem because the amount of CHG applied to skin (10,000 mg/L) is much higher than the minimum inhibitory concentration (MIC) of most Gram-negative bacteria.17 However, it’s worth noting that the concentration of CHG measured on the skin of patients being treated with CHG in one study was considerably lower than the amount applied (15-312 mg/L before the daily bath and 78-1250 mg/L after the daily bath).18 Nonetheless, in this same study, CHG was found to be effective in reducing the skin burden of CRE on patients in a long-term acute care hospital (see Figure, below).18 So, should CHG bathing be applied to combat MDR-GNR?
Figure: Impact of chlorhexidine gluconate (CHG) daily bathing on skin colonization with KPC-producing K. pneumoniae in 64 long-term acute care patients (difference is statistically significant, p=0.01).
A number of studies have implemented CHG as part of a bundle of interventions to control various MDR-GNR. For example, a team from Thailand found that an intervention aimed at improving environmental hygiene combined with CHG brought an outbreak of A. baumannii under control.8 The National Institute of Health Clinical Center19 has included CHG bathing as a component of a successful CRE control bundle, and the same goes for long-term acute care hospitals.6,11 Meanwhile, a Dutch study found that implementing CHG bathing combined with improving hand hygiene failed to reduce the acquisition rate of ESBL Enterobacteriaceae.4 A Mexican study implemented CHG bathing combined with improved hand hygiene and reported a significant reduction in VAP due to A. baumannii.7 However, in all of these studies, it is not possible to tell whether it was the CHG or another element of the bundle that made the difference (or not, in the case of the Dutch study).
No study has been designed specifically to evaluate the impact of CHG daily bathing alone on the rate of Gram-negative bacteria infection or colonization, although rate of Gram-negative bacterial infection or colonization has been reported in several studies of CHG. A small number of high-quality studies that have evaluated CHG as a single intervention including randomization have failed to demonstrate a reduction on Gram-negative BSIs and CLABSIs (see the Climo9, Mlistone10 and Camus16 data from the Table above). Also, a non-randomised before-after study in a trauma ICU reported a non-significant reduction in Acinetobacter species colonization.12 Several other studies mention the rate of Gram-negative infection or colonization in passing, but the numbers are too small for meaningful statistical analysis (see Seyman,5 Popovich13, Bleasdale,14 and Gould15). Although these studies do not provide convincing evidence that CHG works for Gram-negative bacteria, it’s important to remember that they were not powered to evaluate the impact of CHG on Gram-negative bacteria.
One final point to consider is the potential for the development of CHG resistance. Units using CHG universally have reported an increase in the presence of bacteria with reduced CHG susceptibility.20-22 However, the actual degree of reduced susceptibility is moderate, meaning that the clinical importance of this reduced susceptibility is debatable. It is true to say, though, that the potential for meaningful reduced susceptibility is greater in Gram-negative bacteria than in Gram-positive bacteria due to their higher baseline MIC and manifold mechanisms of resistance to biocides and antibiotics.17
So, does CHG bathing work for Gram-negative bacteria? Based on current data, we simply don’t know.
References:
- Derde LP, Dautzenberg MJ, Bonten MJ. Chlorhexidine body washing to control antimicrobial-resistant bacteria in intensive care units: a systematic review. Intensive Care Med 2012; 38: 931-939.
- Vernon MO, Hayden MK, Trick WE et al. Chlorhexidine gluconate to cleanse patients in a medical intensive care unit: the effectiveness of source control to reduce the bioburden of vancomycin-resistant enterococci. Arch Intern Med 2006; 166: 306-312.
- Noto MJ, Domenico HJ, Byrne DW et al. Chlorhexidine Bathing and Health Care-Associated Infections: A Randomized Clinical Trial. JAMA 2015 in press.
- Derde LP, Cooper BS, Goossens H et al. Interventions to reduce colonisation and transmission of antimicrobial-resistant bacteria in intensive care units: an interrupted time series study and cluster randomised trial. Lancet Infect Dis 2014; 14: 31-39.
- Seyman D, Oztoprak N, Berk H, Kizilates F, Emek M. Weekly chlorhexidine douche: does it reduce healthcare-associated bloodstream infections? Scand J Infect Dis 2014; 46: 697-703.
- Hayden MK, Lin MY, Lolans K et al. Prevention of Colonization and Infection by Klebsiella pneumoniae Carbapenemase-Producing Enterobacteriaceae in Long Term Acute Care Hospitals. Clin Infect Dis 2014 in press.
- Martinez-Resendez MF, Garza-Gonzalez E, Mendoza-Olazaran S et al. Impact of daily chlorhexidine baths and hand hygiene compliance on nosocomial infection rates in critically ill patients. Am J Infect Control 2014; 42: 713-717.
- Apisarnthanarak A, Pinitchai U, Warachan B, Warren DK, Khawcharoenporn T, Hayden MK. Effectiveness of infection prevention measures featuring advanced source control and environmental cleaning to limit transmission of extremely-drug resistant Acinetobacter baumannii in a Thai intensive care unit: An analysis before and after extensive flooding. Am J Infect Control 2014; 42: 116-121.
- Climo MW, Yokoe DS, Warren DK et al. Effect of daily chlorhexidine bathing on hospital-acquired infection. N Engl J Med 2013; 368: 533-542.
- Milstone AM, Elward A, Song X et al. Daily chlorhexidine bathing to reduce bacteraemia in critically ill children: a multicentre, cluster-randomised, crossover trial. Lancet 2013; 381: 1099-1106.
- Munoz-Price LS, Hayden MK, Lolans K et al. Successful control of an outbreak of Klebsiella pneumoniae carbapenemase-producing K. pneumoniae at a long-term acute care hospital. Infect Control Hosp Epidemiol 2010; 31: 341-347.
- Evans HL, Dellit TH, Chan J, Nathens AB, Maier RV, Cuschieri J. Effect of chlorhexidine whole-body bathing on hospital-acquired infections among trauma patients. Arch Surg 2010; 145: 240-246.
- Popovich KJ, Hota B, Hayes R, Weinstein RA, Hayden MK. Effectiveness of routine patient cleansing with chlorhexidine gluconate for infection prevention in the medical intensive care unit. Infect Control Hosp Epidemiol 2009; 30: 959-963.
- Bleasdale SC, Trick WE, Gonzalez IM, Lyles RD, Hayden MK, Weinstein RA. Effectiveness of chlorhexidine bathing to reduce catheter-associated bloodstream infections in medical intensive care unit patients. Arch Intern Med 2007; 167: 2073-2079.
- Gould IM, MacKenzie FM, MacLennan G, Pacitti D, Watson EJ, Noble DW. Topical antimicrobials in combination with admission screening and barrier precautions to control endemic methicillin-resistant Staphylococcus aureus in an Intensive Care Unit. Int J Antimicrob Agents 2007; 29: 536-543.
- Camus C, Bellissant E, Sebille V et al. Prevention of acquired infections in intubated patients with the combination of two decontamination regimens. Crit Care Med 2005; 33: 307-314.
- Stickler DJ. Susceptibility of antibiotic-resistant Gram-negative bacteria to biocides: a perspective from the study of catheter biofilms. J Appl Microbiol 2002; 92 Suppl: 163S-170S.
- Lin MY, Lolans K, Blom DW et al. The effectiveness of routine daily chlorhexidine gluconate bathing in reducing Klebsiella pneumoniae carbapenemase-producing Enterobacteriaceae skin burden among long-term acute care hospital patients. Infect Control Hosp Epidemiol 2014; 35: 440-442.
- Palmore TN, Henderson DK. Managing Transmission of Carbapenem-Resistant Enterobacteriaceae in Healthcare Settings: A View From the Trenches. Clin Infect Dis 2013; 57: 1593-1599.
- Horner C, Mawer D, Wilcox M. Reduced susceptibility to chlorhexidine in staphylococci: is it increasing and does it matter? J Antimicrob Chemother 2012; 67: 2547-2559.
- Otter JA, Patel A, Cliff PR, Halligan EP, Tosas O, Edgeworth JD. Selection for qacA carriage in CC22 but not CC30 MRSA bloodstream infection isolates during a successful institutional infection control programme. J Antimicrob Chemother 2013; 68: 992-999.
- Suwantarat N, Carroll KC, Tekle T et al. High prevalence of reduced chlorhexidine susceptibility in organisms causing central line-associated bloodstream infections. Infect Control Hosp Epidemiol 2014; 35: 1183-1186.
Image: John Loo.
Filling the gaps in the guidelines to control resistant Gram-negative bacteria
I gave the third and final installment of a 3-part webinar series on multidrug-resistant Gram-negative rods for 3M recently. You can download my slides here, and access the recording here.
During the webinar, I provided an overview of the available guidelines to control CRE and other resistant Gram-negative bacteria. I then identified gaps in the guidelines, in terms of definitions of standard precautions, outbreak epidemiology and who should be on the guidelines writing dream team. Finally, I discussed some controversial areas in terms of effective interventions: patient isolation, staff cohorting and selective digestive decontamination.
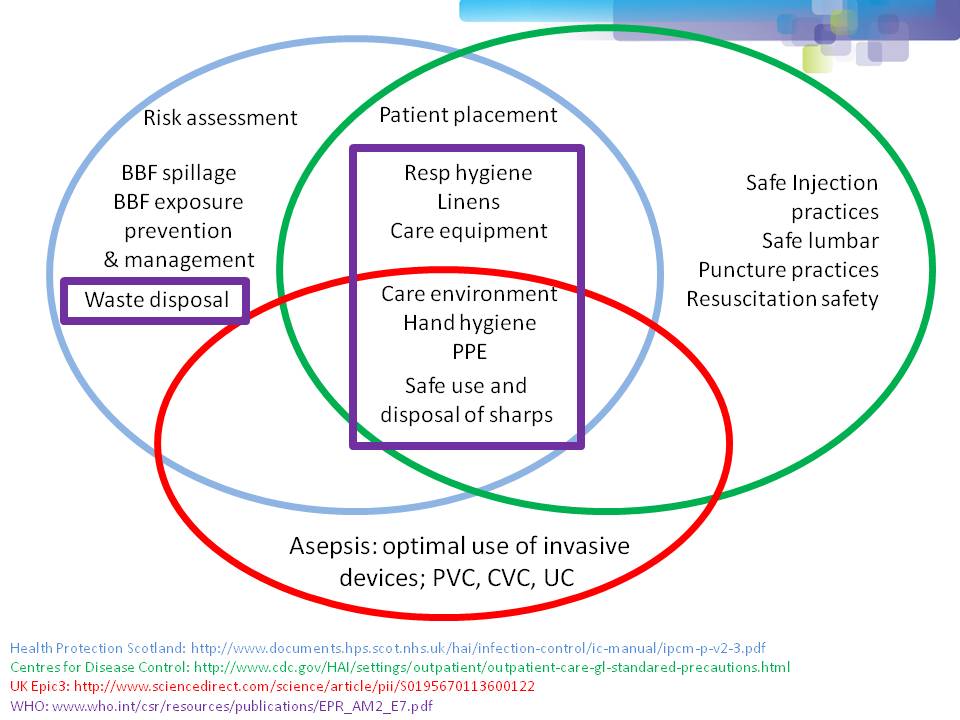
One of the most important points when considering infection prevention and control guidelines is the issue of ‘standard precautions’. What do we apply to every patient, every time? As you can see from Figure 1 below, ‘standard precautions’ is far from standardized. This is problematic when developing and implementing prevention and control guidelines.
Figure 1: differences in the definition of ‘standard precautions’.
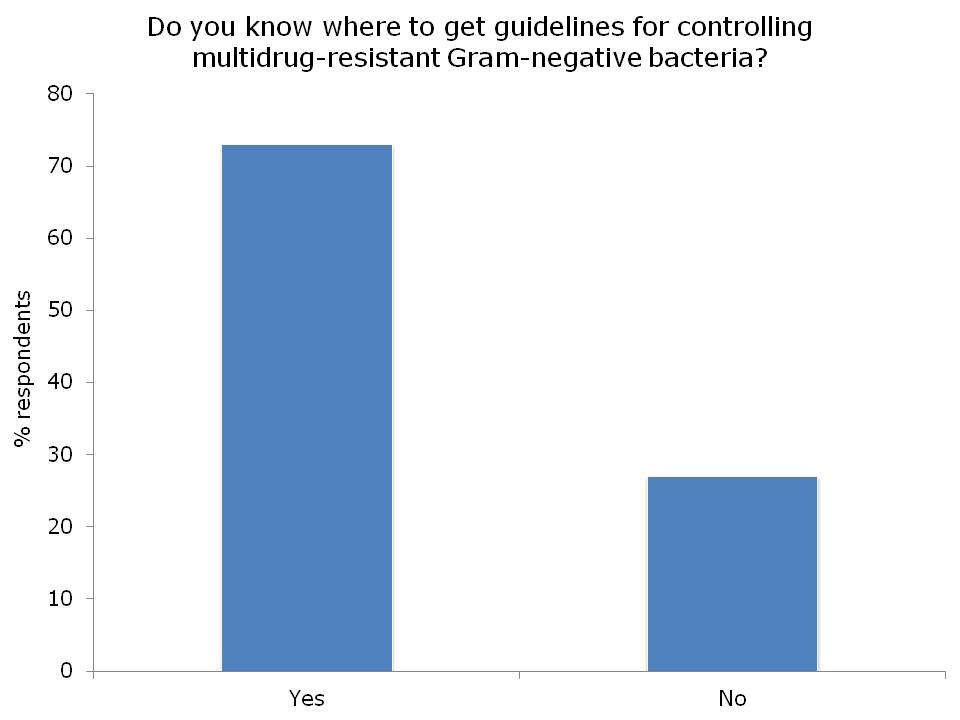
I had the opportunity to ask the webinar audience a few questions throughout the webinar, which are outlined in Figure 2.
Figure 2: response to the questions from the 120 or so participants.
I was somewhat concerned but not that surprised that more than a quarter of the audience did not know where to access control guidelines for MDR-GNR. I suppose this means that we need to do a better job of signposting the location of the various guidelines available. Here’s a non-exhaustive list for starters:
- US CDC CRE Toolkit.
- US AHRQ CRE Toolkit.
- UK Public Health England CPE Tookit.
- UK ESBL guidelines.
- ECDC risk assessment on the spread of spreading (CPE).
- Canadian guidelines for carbapenem resistant GNB.
- Australian recommendations for CRE control.
- ESCMID MDR-GNR control guidelines.
There was a fairly even split between active and passive surveillance to detect outbreaks. The problem with relying on passive surveillance (i.e. clinical cultures) is that there’s a good chance that the ‘horse will have bolted’, and you have a large outbreak on your hands, before a problem is detected. For this reason, I favour active surveillance.
But who to screen? In the case of CRE, I was pleased to see that virtually nobody said nobody. There was a pretty even split between everybody, high-risk individuals or all individuals in high-risk specialties. Accurately identifying individuals who meet screening triggers is operationally challenging, as outlined by the “backlash” to the UK toolkit, so I think screening all patients in high-risk specialties (e.g. ICU) makes most sense.
So, what works to control MDR-GNR transmission? We don’t really know, so are left with a “kitchen sink” (aka bundle approach) (more on this in my recent talk at HIS). We need higher quality studies providing some evidence as to what actually works to control MDR-GNR. Until then, we need to apply a healthy dose of pragmatism!
Being bitten by antibiotic resistant CRAB hurts! (Acinetobacter that is.)
Guest bloggers Dr. Rossana Rosa and Dr. Silvia Munoz-Price (bios below) write…
In everyday practice of those of us who work in intensive care units, a scenario frequently arises: a patient has a surveillance culture growing carbapenem-resistant Acinetobacter baumannii (CRAB). While the ultimate course of action we take will be dictated by the patient’s clinical status, that surveillance culture, in the appropriate context, can provide us with valuable information.
For this study1, we looked at a cohort of patients admitted to a trauma intensive care unit, and sought to identify the risk factors for CRAB infections. We found that patients who had surveillance cultures positive for CRAB had a hazard ratio of 16.3 for the development of clinical infections with this organism, compared to patient’s who remained negative on surveillance, even after adjusting for co-morbidities and antibiotic exposures. Since our results were obtained as part of a well-structured surveillance program, we know that colonization preceded infection. Unfortunately for some of our patients, the time from detection of colonization to development of clinical infections was a matter of days. With therapeutic options for the effective treatment of infections with CRAB limited to tigecycline and polymixins, the consequences of delaying therapy are often fatal. As described by Lee et al, a delay of 48 hour in the administration of adequate therapy for CRAB bacteremia can result in a 50% difference in mortality rate2.
Surveillance cultures are not perfect, and may not detect all colonized patients, but they can be valuable tools in the implementation of infection control strategies3, and as we found in our study, can also potentially serve to guide clinical decision that impact patient care and even survival.
Bio:
Dr. Silvia Munoz-Price (centre left) is an Associate Professor of Clinical Medicine at the Institute for Health and Society, Medical College of Wisconsin, currently serving as the Enterprise Epidemiologist for Froedert & the Medical College of Wisconsin. Dr. Rossana Rosa (centre right) is currently an Infectious Diseases fellow at Jackson Memorial Hospital-University of Miami Miller School of Medicine. She hopes to continue developing her career in Hospital Epidemiology and Infection Control.
References
- Latibeaudiere R, Rosa R, Laowansiri P, Arheart K, Namias N, Munoz-Price LS. Surveillance cultures growing Carbapenem-Resistant Acinetobacter baumannii Predict the Development of Clinical Infections: a Cohort Study. Clin Infect Dis. Oct 28 2014.
- Lee HY, Chen CL, Wu SR, Huang CW, Chiu CH. Risk factors and outcome analysis of Acinetobacter baumannii complex bacteremia in critical patients. Crit Care Med. May 2014;42(5):1081-1088.
- Munoz-Price LS, Quinn JP. Deconstructing the infection control bundles for the containment of carbapenem-resistant Enterobacteriaceae. Curr Opin Infect Dis. Aug 2013;26(4):378-387.
Image: Acinetobacter.