Following hot on the heels of a series of studies showing that daily bathing using chlorhexidine reduces the risk of HCAI, a recent study suggests that chlorhexidine daily bathing does not reduce HCAI. The headline finding is that chlorhexidine bathing did not reduce HCAI. Before throwing out the chlorhexidine with the bathwater, it’s worth considering the limitations of the study.
chlorhexidine
How much chlorhexidine actually makes it to a patient’s skin?
There has been much impressive research of late around the use of chlorhexidine daily bathing to reduce the burden of skin contamination and protect patients from infection. This is quickly becoming en vogue, especially for ICU settings in the USA. But what is compliance like with this intervention, and how do you measure compliance? The studies that have measured compliance previous have said “Yes, this patient was given a chlorhexidine wash today”, but have rarely gone so far as to measure the actual concentration of chlorhexidine on the patient’s skin.
Hot stuff?
 So I’m really quite interested in seasonality of infections. I first became interested in it when looking at increases in E. coli bacteraemia for ARHAI (report here) because of Jennie Wilson’s excellent paper showing seasonality of gram negative bacteraemia, echoed by similar observations and conjecture on warmer weather, more infection. This is true in hospitals as well as the community. Why would this be? We have tussled with increasing E. coli bacteraemia in the UK for a few years now. Goes up every summer, does not return to the baseline, goes up again next summer etc., etc.. Other countries have also reported this. I have heard some suggest this is due to longer hours of daylight leading to more barbeques and more sexual activity. Given that the majority of infections in the UK are >70 years of age, my senior years have no fears for me then.
So I’m really quite interested in seasonality of infections. I first became interested in it when looking at increases in E. coli bacteraemia for ARHAI (report here) because of Jennie Wilson’s excellent paper showing seasonality of gram negative bacteraemia, echoed by similar observations and conjecture on warmer weather, more infection. This is true in hospitals as well as the community. Why would this be? We have tussled with increasing E. coli bacteraemia in the UK for a few years now. Goes up every summer, does not return to the baseline, goes up again next summer etc., etc.. Other countries have also reported this. I have heard some suggest this is due to longer hours of daylight leading to more barbeques and more sexual activity. Given that the majority of infections in the UK are >70 years of age, my senior years have no fears for me then.
Does chlorhexidine bathing work for Gram-negative bacteria?
 The idea of “source control” – using chlorhexidine to reduce the amount of bacteria on a patient’s skin – makes a lot of sense. There’s mounting evidence that chlorhexidine daily bathing works for Gram-positive pathogens, especially in the ICU.1 For example, one of the first thorough studies of chlorhexidine gluconate (CHG) daily bathing showed that the amount of VRE on the skin, in the environment and transmitted to others were all reduced by implementing CHG daily bathing.2 A number of more recent high-quality studies have provided evidence that CHG daily bathing in the ICU setting helps to prevent the transmission of Gram-positive bacteria (see Table below – although note that studies have not been universally positive for CHG).
The idea of “source control” – using chlorhexidine to reduce the amount of bacteria on a patient’s skin – makes a lot of sense. There’s mounting evidence that chlorhexidine daily bathing works for Gram-positive pathogens, especially in the ICU.1 For example, one of the first thorough studies of chlorhexidine gluconate (CHG) daily bathing showed that the amount of VRE on the skin, in the environment and transmitted to others were all reduced by implementing CHG daily bathing.2 A number of more recent high-quality studies have provided evidence that CHG daily bathing in the ICU setting helps to prevent the transmission of Gram-positive bacteria (see Table below – although note that studies have not been universally positive for CHG).
Table: Studies evaluating the impact of chlorhexidine daily bathing (with or without other interventions) including data on Gram-negative bacteria.
| Study | Setting | Design | Intervention | Results |
| Noto 2015 3 | ICU | Cluster RCT | Daily CHG | No significant reduction in HCAI (composite measure including CLABSI, CAUTI, VAP and CDI) |
| Derde 2014 4 | ICU | Time series analysis | Daily CHG plus hand hygiene | Reduction in all MDROs and MRSA (but not VRE or ESBLs) |
| Seyman 2014 5 | ICU | Before-after | Weekly CHG ‘douche’ | Reduction in BSI but not CLABSI; slight reduction in Gram-negative BSI (n too small for statistical analysis) |
| Hayden 2014 6 | LTAC | Before-after | Bundle (including daily CHG) | Acquisition of CRE fell from 4 to 2 per 100 patient weeks |
| Martínez-Reséndez 2014 7 | ICU | Before-after | Daily CHG plus hand hygiene | Reductions in all infections, and in A. baumannii VAP rate |
| Apisarnthanarak 2014 8 | ICU | Before-after | Bundle (including daily CHG) | Reductions in a. baumannii infection and colonization |
| Climo 2013 9 | ICU | Cluster RCT | Daily CHG | Reductions in MRSA / VRE acquisition and all BSI; BSI mainly CoNS (no significant reduction in Gram-negative BSI or CLABSI) |
| Milstone 2013 10 | Paed ICU | Cluster RCT | Daily CHG | BSI reduced; mainly CoNS (no significant reduction in Gram-negative BSI or CLABSI) |
| Munoz-Price 2010 11 | LTAC | Before-after | Bundle (including daily CHG) | CRE carriage prevalence fell from 21% to 0% |
| Evans 2010 12 | ICU | Before-after | Daily CHG | Rate of CLABSI reduced; A. baumannii colonisation reduced but not significantly |
| Popovich 2009 13 | ICU | Before-after | Daily CHG | Rate of CLABSI reduced; A. baumannii infection rate reduced but not significantly |
| Bleasdale 2007 14 | ICU | Cross-over | Daily CHG | Number of Gram-negative BSI in each arm too small for analysis |
| Gould 2007 15 | ICU | Before-after | Daily CHG + mupirocin | Number of Gram-negatives too small for analysis |
| Camus 2005 16 | ICU | RCT | Daily CHG + mupirocin | Number of Gram-negative acquisitions similar in intervention vs. control groups |
The question of whether CHG is effective for the prevention and control of Gram-negative bacteria is rather more complicated. The main issue is that Gram-negative bacteria are less susceptible to CHG than Gram-positive bacteria.17 In theory, this shouldn’t be a problem because the amount of CHG applied to skin (10,000 mg/L) is much higher than the minimum inhibitory concentration (MIC) of most Gram-negative bacteria.17 However, it’s worth noting that the concentration of CHG measured on the skin of patients being treated with CHG in one study was considerably lower than the amount applied (15-312 mg/L before the daily bath and 78-1250 mg/L after the daily bath).18 Nonetheless, in this same study, CHG was found to be effective in reducing the skin burden of CRE on patients in a long-term acute care hospital (see Figure, below).18 So, should CHG bathing be applied to combat MDR-GNR?
Figure: Impact of chlorhexidine gluconate (CHG) daily bathing on skin colonization with KPC-producing K. pneumoniae in 64 long-term acute care patients (difference is statistically significant, p=0.01).
A number of studies have implemented CHG as part of a bundle of interventions to control various MDR-GNR. For example, a team from Thailand found that an intervention aimed at improving environmental hygiene combined with CHG brought an outbreak of A. baumannii under control.8 The National Institute of Health Clinical Center19 has included CHG bathing as a component of a successful CRE control bundle, and the same goes for long-term acute care hospitals.6,11 Meanwhile, a Dutch study found that implementing CHG bathing combined with improving hand hygiene failed to reduce the acquisition rate of ESBL Enterobacteriaceae.4 A Mexican study implemented CHG bathing combined with improved hand hygiene and reported a significant reduction in VAP due to A. baumannii.7 However, in all of these studies, it is not possible to tell whether it was the CHG or another element of the bundle that made the difference (or not, in the case of the Dutch study).
No study has been designed specifically to evaluate the impact of CHG daily bathing alone on the rate of Gram-negative bacteria infection or colonization, although rate of Gram-negative bacterial infection or colonization has been reported in several studies of CHG. A small number of high-quality studies that have evaluated CHG as a single intervention including randomization have failed to demonstrate a reduction on Gram-negative BSIs and CLABSIs (see the Climo9, Mlistone10 and Camus16 data from the Table above). Also, a non-randomised before-after study in a trauma ICU reported a non-significant reduction in Acinetobacter species colonization.12 Several other studies mention the rate of Gram-negative infection or colonization in passing, but the numbers are too small for meaningful statistical analysis (see Seyman,5 Popovich13, Bleasdale,14 and Gould15). Although these studies do not provide convincing evidence that CHG works for Gram-negative bacteria, it’s important to remember that they were not powered to evaluate the impact of CHG on Gram-negative bacteria.
One final point to consider is the potential for the development of CHG resistance. Units using CHG universally have reported an increase in the presence of bacteria with reduced CHG susceptibility.20-22 However, the actual degree of reduced susceptibility is moderate, meaning that the clinical importance of this reduced susceptibility is debatable. It is true to say, though, that the potential for meaningful reduced susceptibility is greater in Gram-negative bacteria than in Gram-positive bacteria due to their higher baseline MIC and manifold mechanisms of resistance to biocides and antibiotics.17
So, does CHG bathing work for Gram-negative bacteria? Based on current data, we simply don’t know.
References:
- Derde LP, Dautzenberg MJ, Bonten MJ. Chlorhexidine body washing to control antimicrobial-resistant bacteria in intensive care units: a systematic review. Intensive Care Med 2012; 38: 931-939.
- Vernon MO, Hayden MK, Trick WE et al. Chlorhexidine gluconate to cleanse patients in a medical intensive care unit: the effectiveness of source control to reduce the bioburden of vancomycin-resistant enterococci. Arch Intern Med 2006; 166: 306-312.
- Noto MJ, Domenico HJ, Byrne DW et al. Chlorhexidine Bathing and Health Care-Associated Infections: A Randomized Clinical Trial. JAMA 2015 in press.
- Derde LP, Cooper BS, Goossens H et al. Interventions to reduce colonisation and transmission of antimicrobial-resistant bacteria in intensive care units: an interrupted time series study and cluster randomised trial. Lancet Infect Dis 2014; 14: 31-39.
- Seyman D, Oztoprak N, Berk H, Kizilates F, Emek M. Weekly chlorhexidine douche: does it reduce healthcare-associated bloodstream infections? Scand J Infect Dis 2014; 46: 697-703.
- Hayden MK, Lin MY, Lolans K et al. Prevention of Colonization and Infection by Klebsiella pneumoniae Carbapenemase-Producing Enterobacteriaceae in Long Term Acute Care Hospitals. Clin Infect Dis 2014 in press.
- Martinez-Resendez MF, Garza-Gonzalez E, Mendoza-Olazaran S et al. Impact of daily chlorhexidine baths and hand hygiene compliance on nosocomial infection rates in critically ill patients. Am J Infect Control 2014; 42: 713-717.
- Apisarnthanarak A, Pinitchai U, Warachan B, Warren DK, Khawcharoenporn T, Hayden MK. Effectiveness of infection prevention measures featuring advanced source control and environmental cleaning to limit transmission of extremely-drug resistant Acinetobacter baumannii in a Thai intensive care unit: An analysis before and after extensive flooding. Am J Infect Control 2014; 42: 116-121.
- Climo MW, Yokoe DS, Warren DK et al. Effect of daily chlorhexidine bathing on hospital-acquired infection. N Engl J Med 2013; 368: 533-542.
- Milstone AM, Elward A, Song X et al. Daily chlorhexidine bathing to reduce bacteraemia in critically ill children: a multicentre, cluster-randomised, crossover trial. Lancet 2013; 381: 1099-1106.
- Munoz-Price LS, Hayden MK, Lolans K et al. Successful control of an outbreak of Klebsiella pneumoniae carbapenemase-producing K. pneumoniae at a long-term acute care hospital. Infect Control Hosp Epidemiol 2010; 31: 341-347.
- Evans HL, Dellit TH, Chan J, Nathens AB, Maier RV, Cuschieri J. Effect of chlorhexidine whole-body bathing on hospital-acquired infections among trauma patients. Arch Surg 2010; 145: 240-246.
- Popovich KJ, Hota B, Hayes R, Weinstein RA, Hayden MK. Effectiveness of routine patient cleansing with chlorhexidine gluconate for infection prevention in the medical intensive care unit. Infect Control Hosp Epidemiol 2009; 30: 959-963.
- Bleasdale SC, Trick WE, Gonzalez IM, Lyles RD, Hayden MK, Weinstein RA. Effectiveness of chlorhexidine bathing to reduce catheter-associated bloodstream infections in medical intensive care unit patients. Arch Intern Med 2007; 167: 2073-2079.
- Gould IM, MacKenzie FM, MacLennan G, Pacitti D, Watson EJ, Noble DW. Topical antimicrobials in combination with admission screening and barrier precautions to control endemic methicillin-resistant Staphylococcus aureus in an Intensive Care Unit. Int J Antimicrob Agents 2007; 29: 536-543.
- Camus C, Bellissant E, Sebille V et al. Prevention of acquired infections in intubated patients with the combination of two decontamination regimens. Crit Care Med 2005; 33: 307-314.
- Stickler DJ. Susceptibility of antibiotic-resistant Gram-negative bacteria to biocides: a perspective from the study of catheter biofilms. J Appl Microbiol 2002; 92 Suppl: 163S-170S.
- Lin MY, Lolans K, Blom DW et al. The effectiveness of routine daily chlorhexidine gluconate bathing in reducing Klebsiella pneumoniae carbapenemase-producing Enterobacteriaceae skin burden among long-term acute care hospital patients. Infect Control Hosp Epidemiol 2014; 35: 440-442.
- Palmore TN, Henderson DK. Managing Transmission of Carbapenem-Resistant Enterobacteriaceae in Healthcare Settings: A View From the Trenches. Clin Infect Dis 2013; 57: 1593-1599.
- Horner C, Mawer D, Wilcox M. Reduced susceptibility to chlorhexidine in staphylococci: is it increasing and does it matter? J Antimicrob Chemother 2012; 67: 2547-2559.
- Otter JA, Patel A, Cliff PR, Halligan EP, Tosas O, Edgeworth JD. Selection for qacA carriage in CC22 but not CC30 MRSA bloodstream infection isolates during a successful institutional infection control programme. J Antimicrob Chemother 2013; 68: 992-999.
- Suwantarat N, Carroll KC, Tekle T et al. High prevalence of reduced chlorhexidine susceptibility in organisms causing central line-associated bloodstream infections. Infect Control Hosp Epidemiol 2014; 35: 1183-1186.
Image: John Loo.
What works to control antibiotic-resistant bacteria in the ICU? A two-for-the-price-of-one study
Not content with a single well-planned study to provide information on what works to control multidrug-resistant organisms (MDROs) in the ICU, the MOSAR study group published an interrupted time series and a cluster randomized trial of various interventions in the Lancet ID. This makes the study rather complex to read and follow, but there are a number of important findings.
Interrupted time series – ‘hygiene’ intervention (chlorhexidine and hand hygiene)
Following a 6-month pre-intervention period, a 6-month interrupted time series of a ‘hygiene’ intervention (universal chlorhexidine bathing combined with hand-hygiene improvement) was performed. The key outcomes were twofold: whether there was a change in trend during each phase, and whether there was a step-change between the phases. The hygiene intervention effected a trend change reduction in all MDROs combined and MRSA individually, but not in VRE or ESBLs (Table). However, there was no step-change compared with the baseline period.
Table: Summary of reduced acquisition of all MDROs combined, or MRSA, VRE and ESBLs individually.
Cluster RCT – screening and isolation
In the 12-month cluster RCT of screening and isolation, the 13 ICUs in 8 European countries were randomized to either rapid screening (PCR for MRSA and VRE plus chromogenic media for ESBL-Enterobacteriaceae) or conventional screening (chromogenic media for MRSA and VRE only). When analysed together, the introduction of rapid or conventional screening was not associated with a trend or step-change reduction in the acquisition of MDROs (Table). In fact, there was an increase in the trend of MRSA acquisition. When comparing rapid with conventional screening, rapid screening was associated with a step-change increase in all MDROs and ESBLs.
Discussion
- The study suggests, prima facie, not to bother with screening and isolation. Indeed, the authors conclude: “In the context of a sustained high level of compliance to hand hygiene and chlorhexidine bathing, screening and isolation of carriers do not reduce acquisition rates of multidrug-resistant bacteria, whether or not screening is done with rapid testing or conventional testing”. However, the major limitation here is that many of the ICUs were already doing screening and isolation during the baseline and hygiene intervention phases! I checked the manuscript carefully (including the supplemental material) to determine exactly how many units were, but it is not disclosed. To make this conclusion, surely the cluster RCT should have been ‘no screening and isolation’ vs. ‘screening and isolation’.
- The increasing trend of MRSA associated with screening and isolation by either method, and step-change increases in all MDROs and ESBLs associated with rapid screening are difficult to interpret. Is an increase in acquisition due to screening and isolation plausible? Can more rapid detection of carriers really increase transmission (the turnaround time was 24 hours for rapid screening, and 48 hours for chromogenic screening)? The rapid screening arm also included chromogenic screening for ESBLs, whereas the conventional screening arm did not, so perhaps this apparent increase in acquisition is due to improved case ascertainment somehow?
- Looking at the supplemental material, a single hospital seemed to contribute the majority of MRSA, with an increasing trend in the baseline period, and a sharp decrease during the hygiene intervention. There’s a suspicion, therefore, that an outbreak in a single ICU influenced the whole study in terms of MRSA. Similarly, a single hospital had a sharp increase in the ESBL rate throughout the screening intervention period, which may explain, to a degree, the increasing trend of ESBL in the rapid screening arm.
- There was an evaluation of length of stay throughout the study phases, with a significant decrease during the hygiene intervention (26%), a significant increase during the rapid screening intervention, and no significant change during the conventional screening intervention. It seems likely that improved sensitivity of rapid screening identified more colonized patients who are more difficult to step down, resulting in an overall increase in length of stay.
- The carriage of qacA and qacB was compared in the baseline and hygiene intervention phase, finding no difference in carriage rate (around 10% for both). This does not match our experience in London, where carriage rates of qacA increased when we introduced universal chlorhexidine bathing. However, this was restricted to a single clone; the acquisition of genes associated with reduced susceptibility to chlorhexidine seems to be clone-specific.
- ICUs varied from open plan to 100% single rooms. Whilst the average proportion of patients in single rooms (15-22%) exceeded the average requirement of patients requiring isolation (around 10%), there was no measure of unit-level variation of single room usage. Since the study was analysed by cluster, the lack of single rooms on some units could have been more important than would appear from looking at the overall average. Put another way, a 100% open plan unit would have been forced to isolate all carriers on the open bay, and vice versa for a 100% single room unit.
- The impact of the various interventions was moderate, even though a ‘high’ MRDO rate was necessary for enrollment (MRSA bacteraemia rate >10%, VRE bacteraemia rate >5%, or ESBL bacteraemia rate >10%). Would the impact of screening and isolation be different on a unit with a lower rate of MDROs? It’s difficult to tell.
- Some of the microbiology is quite interesting: 8% of MRSA were not MRSA and 49% of VRE were not VRE! Also, 29% of the ESBLs were resistant to carbapenems (although it’s not clear how many of these were carbapenemase producers).
In summary, this is an excellent and ambitious study. The lack of impact on ESBL transmission in particular is disappointing, and may lead towards more frequent endogenous transmission for this group. The results do indicate screening and isolation did little to control MDRO transmission in units with improved hand hygiene combined with universal chlorhexidine. However, we need a ‘no screening and isolation’ vs. ‘screening and isolation’ cluster RCT before we ditch screening and isolation.
Perspective from ECCMID 2014 Part I: a voice against ‘selective’ digestive decontamination (SDD)
I enjoyed this year’s ECCMID in Barcelona very much, and came away feeling scientifically, culturally and culinarily enriched! Many thanks to the organizers for such a broad and interesting programme. One of the most interesting sessions was the very final session, on controlling MDROs in the ICU. The session boiled down to the pros and cons of three approaches to decolonization: selective digestive decontamination (SDD), mupirocin for MRSA nasal decolonization, and chlorhexidine gluconate (CHG) bathing. The faculty of Dr Brun-Buisson, Dr Harbarth, Dr Bonten and Dr Huang made it an engaging session.
Selective digestive decontamination (SDD)
The problem is antibiotic resistant bacteria, particularly in the ICU. Is the solution really indiscriminate use of antibiotics to temporarily suppress the load of antibiotic resistant bacteria in the gut? It doesn’t make a lot of sense to me either.
Selective oral decontamination (SOD) or selective digestive decontamination (SDD) is not a new concept, and has been around for some 45 years. Only recently have impressive studies emerged demonstrating that SDD and, to a lesser extent, SOD suppress the load of antibiotic resistant bacteria in the gut, reduce mortality and reduce transmission (de Jonge, de Smet and Daneman). But it’s not without collatoral:
- The use of antibiotics leads to antibiotic resistance, sooner or later. A number of studies suggest that SD is not associated with an increased overall prevalence of resistant bacteria (not least the impressive Daneman review). Indeed, one study showed that rates of resistance actually decreased on units using SD. However, these studies conflate the potential for reducing transmission (and hence reducing unit-level prevalence) with the risk of selecting resistant sub-populations, which both seem likely. One particular concern is the emerging data that SDD drives colistin resistance. Are we playing with fire by overusing our drug of last resort? Furthermore, the abundance of key antibiotic resistance genes doubled on units using SDD when using a microbiotic approach in a recent study.
- Speaking of the microbiome, another speaker described antibiotics as a ‘microbiome-busing atomic bomb’, so perhaps we should rename SDD as ‘scorched earth decontamination’ (SED)! The importance of a happy, healthy microbiota is beginning to dawn on us. We need to make friends with our microbiome, not obliterate it with unindicated antibiotics.
- The impressive studies showing the value of SDD have been performed in the Netherlands, which has a low rate of antibiotic resistance. Will SDD be as effective elsewhere, where the background rates of antibiotic resistance are higher? Indeed, the Daneman study showed a notable (although no statistically significant) increase in the prevalence of MRSA on units using SDD. Is this a case of ‘squeezing the MDRO balloon’?
- SDD temporarily suppresses gut colonization with multidrug-resistant Gram-negative rods but rarely decolonizes permanently. From an infection control viewpoint, it should not change the ‘once positive, always postive’ status quo for resistant Enterobacteriaceae.
I appreciate that I’m presenting a polarized and rather one-side case against SDD here. But for me, whether SDD works is the wrong question: is it the right thing to do? If (perish the thought) I’m a patient in the ICU, then SDD works for me. However, if I’m working on an ICU in 2024 wondering what to do with a pan-drug resistant Gram-negative bacterium, SDD (in 2014) doesn’t work for me.
Mupirocin
Dr Huang presented the key findings from her impressive study of universal CHG combined with MUP. Mupirocin resistance in S. aureus can be low-level mutational) or high-level (acquisition of the Mup resistance genes). The use of MUP has been associated with the development of both high- and low- level resistance. Indeed, several updates from ECCMID show this. For example, Dr Sarah Deeny’s poster showed that low-level resistance appeared to develop during hospitalization. Plus, a study from our group showing that detection of phenotypic high- or low-level mupirocin resistance only represents three quarters of the picture, since carriage of mupirocin resistance determinants remains “silent” 25% of the time.
So, the key question hanging over Dr Huang’s study is the value of universal MUP over and above universal use of CHG. Dr Huang presented an excellent analysis table on this point, which I’ve reproduced below:
Table: Weighing the pros and cons of universal mupirocin use (reproduced with permission from Dr Huang).
One of Dr Huang’s most powerful arguments was that the burden of mupirocin use is in decolonizing a large number of patients prior to elective surgery. Ergo, if you’re going to save MUP, then save it for the highest risk patients (e.g. ICU patients). However, the counter here is that local use of MUP is likely to drive local MUP resistance on the ICU. So, I still feel that we should not recommend the universal use of MUP.
Chlorhexidine
Dr Bonden, Dr Huang and Dr Harbarth reviewed the impressive studies that CHG bathing provides strong protective effects against a range of MDROs (for example Climo, Milstone and Vernon studies). These studies are not without their critics – some say that the effect on reducing relatively benign coagulase-negative staphylococci BSI amplifies the overall effect. However, both the data and rationale are stong: if you reduce the amount of MDRO on the patients’ skin (‘source control’), you reduce the chances of endogeneous infection, and transmission to others. Unlike antibiotics, CHG is a biocide with a less specific molecular target, which makes resistance more challenging from a bacterial viewpoint. However, reduced susceptibility to chlorhexidine must be monitored carefully. A number of studies have hinted that reduced susceptibility to chlorhexide may be an emerging problem, (for example Batra, Otter and Lee.) But increases in bacterial MICs (for Gram-positive bacteria at least) appear to be a long way below the applied concentration. However, it’s worth noting that the measured CHG skin concentration in one study (15-312 mg/L before the daily bath and 78-1250 mg/L after the daily bath) was much lower than the applied CHG concentration (10,000 mg/L). This is around the CHG MIC for some Gram-negatives and potentially brings the subtly reduced susceptibility to CHG reported in MRSA into play. On balance though, the rationale and data on reduced susceptibility are cautionary but not enough to recommend against universal use in the ICU given the clinical upside.
Conclusion
What shoud be the standard of care for ICU patients? My current view is: universal CHG, targeted mupirocin for MRSA decolonization and absolutely no SDD!
Headlines from ECCMID
I’ll be posting some blogs on some of these topics over the coming days. You can view some other ‘Perspectives from ECCMID’ here.
- We are still no closer to figuring out what works to control multidrug resistant Gram-negative rods (including CRE).
- CDI does not seem to be emerging as a community pathogen, despite apparent increases.
- Bacteriotherpy for synthetic faecal microbiota transplant (FMT, aka transpoosion) is getting close.
- We need to stop polluting our plant by pumping antibiotics into our environment.
- As one tweeter (@marina_manrique) put it, whole genome sequencing (WGS) has becoming a bit like the One Ring from Tolkein’s Lord of the Rings: ‘one ring to rule them all, one ring to bind them, one ring to bring them all, and in the outbreak find them (out – the other methods that is)’.
Picture credit: ‘Antibiotics’.
Universal MRSA decolonization will tend towards universal resistance
 A seminal study recently published in the New England Journal of Medicine evaluated the impact of universal decolonization vs. targeted screening and / or decolonization to prevent ICU infection. The study has the potential to fundamentally change practice in ICUs around the world. However, I have two major problems with the study, namely mupirocin and chlorhexidine.
A seminal study recently published in the New England Journal of Medicine evaluated the impact of universal decolonization vs. targeted screening and / or decolonization to prevent ICU infection. The study has the potential to fundamentally change practice in ICUs around the world. However, I have two major problems with the study, namely mupirocin and chlorhexidine.
Study design and results. The authors describe their design as a ‘pragmatic’ randomized controlled trial, which should serve as a model for other studies. There were several variables that the authors could not change, for example, the fact that some US states mandate universal screening whereas others do not. They were able to cleverly incorporate this into their stratified randomization process so that the integrity of the study was not compromised.
The study was large, with 74,256 patients admitted to 74 ICUs in 43 hospitals randomized to the following groups during the intervention phase:
Group 1: Targeted screening and isolation.
Group 2: Targeted screening, isolation and decolonization (using intranasal mupirocin and chlorhexidine bathing).
Group 3: Universal decolonization (using intranasal mupirocin and chlorhexidine bathing).
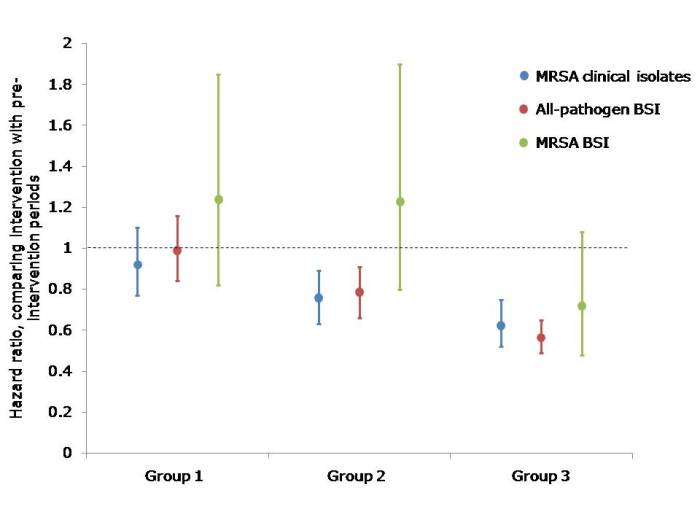
The study compared the hazard ratio for MRSA clinical cultures, all-pathogen bloodstream infections and MRSA bloodstream infections in the 12 month pre-intervention vs. the 18 month intervention period for each of the groups. The main finding was that universal decolonization (Group 3) was associated with significantly lower rates of MRSA clinical cultures and all-pathogen bloodstream infection, and reduced rates of MRSA bloodstream infection (Chart). Target screening and isolation (Group 1) was the least effective strategy and targeted screening, isolation and decolonization (Group 2) had intermediate effectiveness (Chart).
[Chart: hazard ratios and 95% confidence intervals comparing the pre-intervention with the intervention period in each of the three groups. Group 1: Targeted screening and isolation; Group 2: Targeted screening, isolation and decolonization; Group 3: Universal decolonization.]
Major problems: My major issue with the study is the universal (aka indiscriminate) use of mupirocin and, to a lesser extent, chlorhexidine. MRSA are able to acquire low-level resistance to mupirocin through mutations in ileS or high-level resistance through the acquisition of mupA. I can’t help thinking that the authors underestimate the risk of driving mupirocin resistance with their comment in the discussion: ‘Mupirocin resistance has been reported in some studies of MRSA decolonization, but not all such studies.’ It is frankly naive to think that universal use of mupirocin for each ICU admission will not result in the widespread development of mupriocin resistance. Thus, the comment in the accompanying editorial urging caution in implementing the universal use of mupirocin is warranted.
The situation is not as clear-cut with chlorhexidine. Clinically significant reduced susceptibility to chlorhexidine in S. aureus has been hard to define for a number of reasons:
- There is no generally agreed methodology for detecting reduced chlorhexidine susceptibility.
- Population MICs vary considerably making a meaningful breakpoint difficult to determine.
- Higher MICs displayed by some isolates (4–16 mg/L) are still well below the effective chlorhexidine concentration applied to skin (10000–40000 mg/L).
- Although genes encoding membrane efflux pumps have been identified in S. aureus, including qacA, these have an unclear relationship with reduced susceptibility.
- Other mechanisms of reduced susceptibility, such as decreased uptake of chlorhexidine, may confound the apparent effect of efflux pumps.
- Although many studies have reported on qacA carriage or reduced chlorhexidine susceptibility in S. aureus isolates, few have evaluated clinically relevant outcomes.
Notwithstanding these limitations, it is clear that the implementation of chlorhexidine based decolonization drives an increase in the carriage of genes associated with reduced susceptibility to chlorhexidine, and this may be clinically relevant based on studies from London and Geneva. For example, a recent article from my research lab in London found that the implementation of universal chlorhexidine-based decolonization was associated with the selection of qacA linked with a higher chlorhexidine MIC in one dominant endemic MRSA clone (CC22), but not another (CC30). The slower reduction in the CC22 MRSA bloodstream infection rate suggests that carriage of qacA confers a selective advantage.
So, will the introduction of universal decolonization result in resistance to mupirocin and chlorhexidine? The answer for mupirocin is a resounding yes; the answer for chlorhexidine is currently not clear.
Other limitations: It’s always easy to snipe at a high-profile study, but there do seem to be some important limitations. First, nasal screens alone were performed in patients admitted to Groups 1 and 2. The sensitivity of nasal screens alone can be as low as 50%, so the inclusion of other screening sites are preferable to improve sensitivity. The implications of missing many carriers on admission in Groups 1 and 2 are obvious.
Second, although the trend was in the same direction, there was no significant reduction on MRSA bloodstream infections for patients in Group 3. In fact, the crude rate of MRSA bloodstream infection was rather high in all three groups (0.5 to 0.7 per 1000 patient days in all groups during the baseline and intervention periods). For reference, the rate of MRSA bloodstream infection at Guy’s and St. Thomas’ NHS Foundation Trust in London was lower than this at the peak of the MRSA problems in 2003, approximately 10 times lower by 2008 and has fallen further since then. So, why the relatively high rate of MRSA bloodstream infection across the board in this study?
Third, while randomization is the “gold standard” in terms of study design, by chance, three of four hospitals that performed bone marrow and solid organ transplantation ended up in Group 3, which skewed this population somewhat. The study was large enough to “smooth out” this skew, but small studies that attempt randomization should take note. In a recent study with Johns Hopkins, we only had 6 units to play with so decided not to randomize but instead roughly “matched” the units in the two arms, which may be a better approach in smaller studies.
Fourth, it does not seem that any attempt was made to ensure that patients in Groups 1 and 2 were bathed daily. While bathing with chlorhexidine impregnated cloths has been shown to be superior to bathing with soap and water, it is not possible to determine whether daily bathing or chlorhexidine were most important.
Finally, compliance will never be 100%. Only 81% of patients in Group 3 actually received chlorhexidine and 86% received mupriocin, and only approximately 90% of patients in Group 2 that were due decolonization received chlorhexidine and mupriocin. However, the authors did achieve a very high compliance (in the high 90s) with admission screening in Groups 1 and 2.
Summary: The authors should be complimented for performing such a detailed and useful study. Unfortunately, the protocol was not configured to assess the relative contribution of mupirocin and chlorhexidine to the reductions in infection. Given the likely emergence of mupirocin resistance, future studies should drop the universal use of mupirocin all together and see how far universal chlorhexidine alone can go; recent data suggest it will go a long way. Indeed, the UK has managed to make a dramatic, national reduction in the rate of MRSA bloodstream infection without implementing universal mupirocin.
I have grave concerns about the universal use of mupirocin for all ICU admissions and, perish the thought, for all hospital admissions. To decide on implementing universal mupriocin use with a plan to keep an eye on resistance rates seems remiss (to put it politely) since the likelihood of widespread resistance is so high. I am less concerned about the universal use of chlorhexidine, although reduced susceptibility needs to be monitored carefully.
The accompanying editorial was entitled ‘Screening inpatients for MRSA – the case is closed’. For me, the only closed case emerging from this study is that universal, indiscriminate use of mupriocin is a bad idea. Antibiotics have caused the problem; they are not the solution.
Article citation: Huang et al. Targeted versus universal decolonization to prevent ICU infection. New Engl J Med 2013 May 29. [Epub ahead of print].