You may have seen some commentary and debate on a recently updated Cochrane review on physical interventions to interrupt the spread of respiratory viruses. I’m stepping into the debate only to clarify a few small points – and to provide an overview of what the review does (and does not) tell us. However, my firm advice is to read the review for yourself and come to your own conclusions.
Continue readingcluster RCT
Single rooms for ESBLs anyone?
A comprehensive and impressive cluster randomised crossover study published in Lancet ID examines whether it makes sense to use single rooms (as compared with multi-bed bays) to apply contact precautions for patients known to be carrying ESBL-Enterobacteriaceae. I need to be careful what I say, because fellow bloggers Marc and Andreas are co-authors. However, the gist seems to be: don’t bother with single rooms for ESBL-E carriers – but many hospitals don’t have capacity to do that anyway, so this may not be a practice-changing finding in many parts of the world!
Time to go shopping for a UVC system?
It is great to see the long-awaited ‘Benefits of Terminal Room Disinfection’ (BETR-D) randomised controlled trial of a UVC automated room decon (ARD) system published, in the Lancet, no less! This study firms up the importance of environmental contamination in transmission, and demonstrates additional benefit of UVC over and above enhanced conventional methods for VRE, maybe for MRSA, but not for C. difficile.
How can we stop nursing homes nurturing MRSA?
There is an emerging feeling that we need to start spreading the focus of infection prevention and control beyond acute hospitals. There has always been a sense that standards of infection control outside of acute settings are, shall we say, “different” to acute hospitals (aka non-existent) so it’s great to see a study of an infection control intervention in nursing homes.
The study was a cluster randomised controlled trial of MRSA screening, decolonisation and enhanced environmental disinfection vs. standard precautions in 104 of 157 nursing homes in a Swiss region. The authors chose a rather unusual, pragmatic endpoint of the prevalence of MRSA colonisation after 12 months.
What works to control antibiotic-resistant bacteria in the ICU? A two-for-the-price-of-one study
Not content with a single well-planned study to provide information on what works to control multidrug-resistant organisms (MDROs) in the ICU, the MOSAR study group published an interrupted time series and a cluster randomized trial of various interventions in the Lancet ID. This makes the study rather complex to read and follow, but there are a number of important findings.
Interrupted time series – ‘hygiene’ intervention (chlorhexidine and hand hygiene)
Following a 6-month pre-intervention period, a 6-month interrupted time series of a ‘hygiene’ intervention (universal chlorhexidine bathing combined with hand-hygiene improvement) was performed. The key outcomes were twofold: whether there was a change in trend during each phase, and whether there was a step-change between the phases. The hygiene intervention effected a trend change reduction in all MDROs combined and MRSA individually, but not in VRE or ESBLs (Table). However, there was no step-change compared with the baseline period.
Table: Summary of reduced acquisition of all MDROs combined, or MRSA, VRE and ESBLs individually.
Cluster RCT – screening and isolation
In the 12-month cluster RCT of screening and isolation, the 13 ICUs in 8 European countries were randomized to either rapid screening (PCR for MRSA and VRE plus chromogenic media for ESBL-Enterobacteriaceae) or conventional screening (chromogenic media for MRSA and VRE only). When analysed together, the introduction of rapid or conventional screening was not associated with a trend or step-change reduction in the acquisition of MDROs (Table). In fact, there was an increase in the trend of MRSA acquisition. When comparing rapid with conventional screening, rapid screening was associated with a step-change increase in all MDROs and ESBLs.
Discussion
- The study suggests, prima facie, not to bother with screening and isolation. Indeed, the authors conclude: “In the context of a sustained high level of compliance to hand hygiene and chlorhexidine bathing, screening and isolation of carriers do not reduce acquisition rates of multidrug-resistant bacteria, whether or not screening is done with rapid testing or conventional testing”. However, the major limitation here is that many of the ICUs were already doing screening and isolation during the baseline and hygiene intervention phases! I checked the manuscript carefully (including the supplemental material) to determine exactly how many units were, but it is not disclosed. To make this conclusion, surely the cluster RCT should have been ‘no screening and isolation’ vs. ‘screening and isolation’.
- The increasing trend of MRSA associated with screening and isolation by either method, and step-change increases in all MDROs and ESBLs associated with rapid screening are difficult to interpret. Is an increase in acquisition due to screening and isolation plausible? Can more rapid detection of carriers really increase transmission (the turnaround time was 24 hours for rapid screening, and 48 hours for chromogenic screening)? The rapid screening arm also included chromogenic screening for ESBLs, whereas the conventional screening arm did not, so perhaps this apparent increase in acquisition is due to improved case ascertainment somehow?
- Looking at the supplemental material, a single hospital seemed to contribute the majority of MRSA, with an increasing trend in the baseline period, and a sharp decrease during the hygiene intervention. There’s a suspicion, therefore, that an outbreak in a single ICU influenced the whole study in terms of MRSA. Similarly, a single hospital had a sharp increase in the ESBL rate throughout the screening intervention period, which may explain, to a degree, the increasing trend of ESBL in the rapid screening arm.
- There was an evaluation of length of stay throughout the study phases, with a significant decrease during the hygiene intervention (26%), a significant increase during the rapid screening intervention, and no significant change during the conventional screening intervention. It seems likely that improved sensitivity of rapid screening identified more colonized patients who are more difficult to step down, resulting in an overall increase in length of stay.
- The carriage of qacA and qacB was compared in the baseline and hygiene intervention phase, finding no difference in carriage rate (around 10% for both). This does not match our experience in London, where carriage rates of qacA increased when we introduced universal chlorhexidine bathing. However, this was restricted to a single clone; the acquisition of genes associated with reduced susceptibility to chlorhexidine seems to be clone-specific.
- ICUs varied from open plan to 100% single rooms. Whilst the average proportion of patients in single rooms (15-22%) exceeded the average requirement of patients requiring isolation (around 10%), there was no measure of unit-level variation of single room usage. Since the study was analysed by cluster, the lack of single rooms on some units could have been more important than would appear from looking at the overall average. Put another way, a 100% open plan unit would have been forced to isolate all carriers on the open bay, and vice versa for a 100% single room unit.
- The impact of the various interventions was moderate, even though a ‘high’ MRDO rate was necessary for enrollment (MRSA bacteraemia rate >10%, VRE bacteraemia rate >5%, or ESBL bacteraemia rate >10%). Would the impact of screening and isolation be different on a unit with a lower rate of MDROs? It’s difficult to tell.
- Some of the microbiology is quite interesting: 8% of MRSA were not MRSA and 49% of VRE were not VRE! Also, 29% of the ESBLs were resistant to carbapenems (although it’s not clear how many of these were carbapenemase producers).
In summary, this is an excellent and ambitious study. The lack of impact on ESBL transmission in particular is disappointing, and may lead towards more frequent endogenous transmission for this group. The results do indicate screening and isolation did little to control MDRO transmission in units with improved hand hygiene combined with universal chlorhexidine. However, we need a ‘no screening and isolation’ vs. ‘screening and isolation’ cluster RCT before we ditch screening and isolation.
This study has been BUGGing me for a while
A fabulous study recently published in JAMA evaluates the ‘Benefits of Universal Glove and Gown’ (BUGG) in US ICUs. This is a model study design: one of the first cluster randomized controlled trials of a non-therapeutic infection control intervention. Twenty ICUs were paired and randomized to either universal glove and gowning, or to continue the current practice of placing patients known to be infected or colonized with MRSA and VRE on contact precautions. The hypothesis is that undetected colonization with MRSA and VRE is common, and the only real way to address this is to assume everybody is colonized!
Summary of findings:
- Universal glove and gowning was not associated with a reduction in a composite measure of MRSA / VRE acquisition (the primary outcome).
- VRE acquisition was not reduced by universal glove and gown use, whereas MRSA was.
- CLABSI, CAUTI and VAP; ICU mortality; and adverse events did differ significantly between the two groups.
- Hand hygiene compliance on room entry was not significantly different between the two arms, whereas hand hygiene compliance on room exit was significantly higher in the intervention arm.
- Healthcare workers visited patients 20% less frequently in the intervention arm (4.2 vs. 5.2 visits per hour).
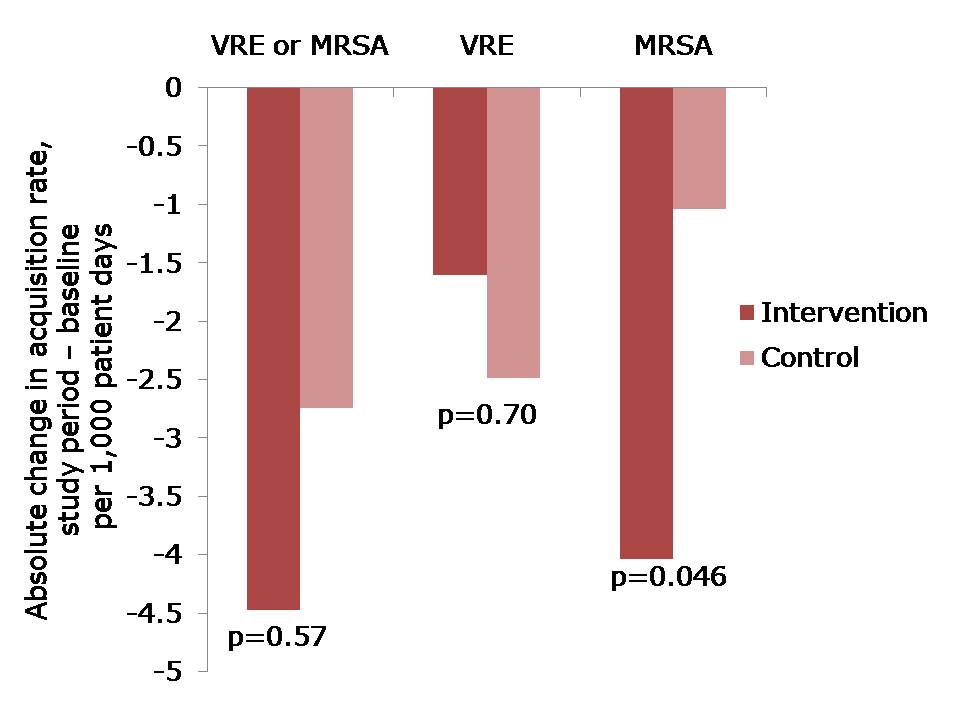 Figure: The change in acquisition rate, comparing the baseline period with the study period for the intervention and control units.
Figure: The change in acquisition rate, comparing the baseline period with the study period for the intervention and control units.
Here’s what’s BUGGing me about this study:
- The acquisition rate in both intervention and control study arms reduced (Figure). The acquisition rate reduction in the control arms may be due to improved compliance with admission screening, resulting in more accurate ascertainment of who required contact precautions.
- The significant reduction was achieved for MRSA but not for VRE. The authors suggest that VRE colonization may have been suppressed on admission and not detected, and flourished during antimicrobial therapy giving the impressive of acquisition. I wonder whether differences in the routes of transmission may also have contributed; for example, VRE seems to be substantially “more environmental” than MRSA. Another potential confounder is that, by chance, the prevalence of MRSA or VRE on admission to the intervention ICUs was more than double that in the control ICUs (22% vs. 9%). In actual fact, the raw rate of MRSA acquisition in the intervention ICUs was marginally higher than in the control ICUs during the intervention period (6.00 vs. 5.94 per 1000 patient days), even though the change in rate was significantly greater on the intervention ICU. Although adjustment was made for this difference in the analysis, it may have skewed the findings somewhat.
- The authors achieved remarkably high compliance with admission screening (around 95%), discharge screening (around 85%) and glove and gowning (around 85%). Each site had the luxury of a study coordinator and a physician champion to lead implementation, plus weekly feedback on screening compliance and visits from study investigators. Most ICUs would not be afforded these luxuries so I suspect that real-world compliance outside of the somewhat artificial study environment would be considerably lower. Indeed, an ID Week poster suggests that compliance with gowning in one US ICU was a ‘dismal’ 20%!
- Adverse events were not significantly higher in the universal glove and gowning arm, which may seem surprising prima facie. However, the reason why adverse events are more common for patients on contact precautions is that they are marginalized by being on contact precautions. If all patients are effectively on contact precautions, the time of healthcare workers would be spread evenly.
- Universal gloving is likely to result in universally bad hand hygiene compliance within the room during patient care; when healthcare workers feel protected, they are less likely to comply with hand hygiene and gloves are a good way to make healthcare workers feel protected. The increase in hand hygiene compliance on room exit is probably also a symptom of inherent human factors, since healthcare workers feel more ‘dirty’ when exiting the room of a patient with a higher perceived risk of MDRO ‘contamination’ (the so-called “urgh” factor).
- Healthcare workers had less time for patient care in the intervention arm because they were busy donning and doffing gloves and gowns. Interestingly, the authors suggest that fewer visits may be a good thing for patients, and may have contributed to their reduced chances of acquiring MRSA. This seems unlikely though, given the fact that VRE acquisition was not reduced. On balance, less contact with healthcare workers is likely to be bad for patients.
- The increased cost of universal glove and gowning was not evaluated and, whilst incrementally small, would be a substantial sum.
In summary, this study sets the standard in terms of rigorous assessment of an infection prevention and control intervention. Universal application of gloves and gowns is unlikely to do as much harm as universal administration of mupirocin, but it will not make a profound reduction in the transmission of MDROs. Therefore, I shouldn’t think many ICUs will be rushing to implement universal gloves and gowns on the strength of these findings.


