It is great to see the long-awaited ‘Benefits of Terminal Room Disinfection’ (BETR-D) randomised controlled trial of a UVC automated room decon (ARD) system published, in the Lancet, no less! This study firms up the importance of environmental contamination in transmission, and demonstrates additional benefit of UVC over and above enhanced conventional methods for VRE, maybe for MRSA, but not for C. difficile.
Clostridium difficile
Should we start admission screening for C. difficile carriage? A Kiernan vs. Otter pro-con debate!
Both Martin and Jon wanted to post a blog about the same article, so thought we’d put our hands together, so to speak, into a pro-con format! We hope you find it useful.
Reflections from ECCMID: the pink, the blue, and the new
I’ve recently returned from an enjoyable few days in Amsterdam for ECCMID. I’ve not been to a conference of this scale for a few years; there was a lot of good stuff to choose from so I’ve tried to stick to key updates for the purposes of this reflection!
English HCAI and AMR data at your Fingertips
PHE have just launched an interactive database for HCAI and AMR data from England using their ‘Fingertips’ platform. The HCAI data in the module has been available for years, but was buried in pretty dense Excel spreadsheets so tricky to visualise. The Fingertips platform makes data extraction and visualisation so easy even I can do it (example below).
Reflections from HIS 2014, Part II: Dealing with the contaminated environment
Welcome to Part II of my reflections from HIS. For the box-set, see the list at the beginning of Part I here.
Dr Karen Vickery – Multispecies biofilms on dry hospital surfaces – harbouring and protecting multiantibiotic resistant organisms
Probably the most important update from the entire conference was more data from the Vickery lab on biofilms on dry hospital surfaces. She excised 44 dry surface samples from the ICU, put them under the electron microscope and, lo and behold, 41 of them (93%) had fully-fledged (if somewhat unusual) EPS-producing biofilms on! The implications are huge: this could explain extended surface survival, poor success rate of surface sampling, and result in reduced biocide susceptibility up to the tune of 1000x (see my review just published in JHI with Karen as a co-author for more on biocides and biofilm susceptibility).
Dr Silvia Munoz-Price – Controlling multidrug resistant Gram-negative bacilli in your hospital: We can do it so can you!
Dr Munoz-Price described her hospital’s impressive reductions on carbapenem-resistant A. baumannii – from 12 new isolates per week to virtually none today. So what worked? It’s difficult to be sure since it was a bundled intervention. Dr Munoz-Price described the rationale behind some elements of the bundle: environmental surface and staff hand sampling to visualize the invisible, environmental cleaning and disinfection to deal with the ‘fecal [sic] patina’ [a stooly veneer emanating from the rectum] (see Dr Munoz-Price and Dr Rosa’s guest blog for more details), and chlorhexidine bathing. Perhaps the most interesting aspect was the various implementation challenges that were overcome. It was amazing how far removed practice ‘in the trenches’ was from the policy set by the epidemiologist’s office, exemplified by environmental staff buying their own UV lamps to for “spot cleaning” removal of fluorescent markers of cleaning thoroughness. Overcoming these challenges required more that the stick (citations for non-compliance, which failed); culture change takes understanding, time and a very large carrot (and some sticks too, sometimes).
Jim Gauthier – faeces management
A number of key pathogens are associated with faecal colonization and shedding: C. difficile, VRE, ESBL and CRE. Jim didn’t mention MRSA, but this can also cause gastrointestinal colonization and, more controversially, infection. Enterobacteriaceae can survive on dry surfaces for longer than you’d expect, too. We traditionally worry about surface contamination of high-touch sites in inpatient settings. Floor contamination isn’t important (unless you happen to be a wheel chair user, a toddler, or drop your pen). Contamination in outpatient settings isn’t a problem either (unless you happen to have a fairly short consultation for a patient with VRE). So, what to do? Jim introduced the idea of a ‘hierarchy of control’; put another way, prevention is better than cure, so do we have the right systems in place to manage faeces which is teeming with hospital pathogens? For example, should we be enforcing mandatory contact precautions for all contact with faeces (standard precautions – which aren’t very standard anyway – are probably not adequate)? Finally, Jim mentioned the growing importance of faecal microbiota transplantation (and hearing a Canadian speak about this reminded me of a hilarious spoof video).
No-touch automated room decontamination (NTD)
 Figure: Hospital bed rails are frequently contaminated, and often not easy to clean and disinfect using conventional methods.
Figure: Hospital bed rails are frequently contaminated, and often not easy to clean and disinfect using conventional methods.
Paul Dickens – establishing Ebola surge isolation capacity in the UK
Paul Dickens gave a whistle-stop overview of the detailed plans for Ebola surge capacity in the UK (perish the thought). He began by describing the replacement of formaldehyde with hydrogen peroxide vapour for the decontamination of the patient isolators at the Royal Free High Level Isolation Unit (HLIU). They now have a tried and tested process and protocols in place to get the HLIU back online within days using hydrogen peroxide vapour decontamination, where the previous protocol using formaldehyde put it out of action for 6 weeks! (I was involved in writing the protocols for this tricky decontamination assignment, which were reported on a poster published at HIS.) Other challenges in establishing surge capacity include staff expertise, and PPE recommendations, supply & training. Surge capacity is now established. Let’s just hope we won’t need it!
Dr Frédéric Barbut – How to eradicate Clostridium difficile spores from the environment
There’s now plenty of evidence that contaminated surfaces contribute to the transmission of C. difficile. These environmental intervention studies show a 50-80% reduction in the rate of CDI; does this mean that 50-80% of CDI acquisition is environmentally-associated? This seems too high, but it’s difficult to think of another explanation. Furthermore, there is emerging but compelling evidence of a proportional relationship between the degree of C. difficile surface contamination and transmission risk? I really don’t think that the public have yet ‘got’ that the previous occupant can influence acquisition risk. And when they do, I think there will be increasing demand for properly decontamination rooms. So, is it time to turn to NTD systems? Sometimes, yes. And do you go for hydrogen peroxide or UV? Well, that depends on what you’re trying to achieve! If you’re trying to eliminate pathogens, which sometimes you will be, then hydrogen peroxide vapour is the best choice. But if you’re trying to reduce contamination levels without necessarily eliminating all pathogens, then UV is the best choice due to its speed and ease of use.
The debate: “Hospitals that do not use high-tech decontamination of the environment are doing their patients a disservice.”
This debate pitted Profs Hilary Humphreys and Phil Carling (pro) against Peter Hoffman and Martin Kiernan (con). It was lively, entertaining and engaging…
Prof Humphreys argued that it is not acceptable to admit patients to rooms with inherent additional risk for transmission. We can address this by ‘walking like the Egyptians’ and copperising our surfaces, for which there is now some data with a clinical outcome. Another approach is NTD systems, for which data (including some clinical outcomes) are emerging. Prof Carling’s presentation was somewhat unusual, with his arguments seemingly an appeal to common sense rather than drawn from the published literature.
Martin Kiernan began by acknowledging the role of the environment, but that hand contamination is almost always the final vector (and there’s some evidence for this). The cornerstone of Martin’s argument was that whether NTD systems work is the wrong question. We should be focusing our time, money and attention on improving conventional methods which have been shown to reduce transmission. Peter Hoffman complemented Martin’s pragmatic viewpoint with thorough, thoughtful critiques of the studies on HPV decontamination with a clinical outcome. The 2008 Boyce study has more holes than the 2013 Passaretti study, which itself is far from watertight!
The key argument for turning to NTD systems is that admission to a room previously occupied by a patient with an MDRO increases the risk of acquisition due to residual contamination, and NTD decontamination mitigates this increased risk. So, my own conclusion is that hospitals that do not use high-tech decontamination of the environment are indeed doing their patients a disservice. Sometimes!
Look out for the third and final installment of my reflections from HIS 2014 at some point tomorrow!
APIC New England 2014 Conference Report: ‘The how, when, & where of C. diff – can you “C” the difference?’
I was privileged to be asked to speak at the APIC New England Conference today in Springfield, Massachusetts. It was a vibrant day, and congratulations to the organizing committee for putting on such an enjoyable event.
Marie-Louise Landry MD – Continuing conundrums and controversies in the laboratory diagnosis of Clostridium difficile.
Dr Landy, a virologist by trade, began by reflecting on the fact that CDI is relatively new phenomenon, discovered in the late 1970s and initially thought to be viral! Having briefly presented the clinical problem and pathogenesis of CDI, Dr Landry got to the nitty gritty of how to test for CDI. You can choose to target the toxin, bacteria or bacteria capable of producing toxin:
- Toxin: cytotoxicity cell culture assay (complex and requires overnight incubation) or enzyme immuno assay (terrible sensitivity).
- Bacteria: culture (slow and doesn’t tell you much) or rapid GDH antigen assay (sensitive, but requires a confirmatory test of toxigenicity).
- Bacteria capable of producing toxin: toxigenic culture (requires incubation) or Nucleic Acid Amplification Test (NAAT) such as PCR or LAMP (rapid, expensive).
To be honest, before Dr Landry’s talk, I thought that testing for CDI was pretty much sorted: GDH as a sensitive screening test following by PCR to detect the toxin gene for GDH positives. However, Dr Landry presented a compelling case that whilst GDH makes sense as a screening test, detecting the toxin gene via PCR is only half the story: the real gold-standard test is a cytotoxicity cell culture assay to confirm that the disease-causing toxin is present. Cost & clinical association makes compelling case for cell culture cytotoxicity assays; convenience for PCR! Indeed is the initial enthusiasm for PCR CDI testing waning as reality sets in (like the famous ‘Going and Coming’ by Rockwell)?
A final point for discussion: you can have the best laboratory diagnostics in the world, but if you’re testing inappropriate specimens, you’ll end up with false positives. We need a firm “no diarrhea, no CDI diagnostics” rule!
Curtis Donskey MD – Controlling the spread of C. difficile: a multifaceted approach
Dr Donskey began by considering that no healthcare facility is an island, and that long-term care facilities are an integral part of CDI spread. Dr Donskey spent most of the talk considering the environmental considerations related to CDI. Why does cleaning fail? Due to poor implementation: a research team with a bucket of bleach can eliminate C. difficile from surfaces! Various tools are available to help us tackle C. difficile environmental contamination. However, fluorescent markers and UVC did not eliminate C. difficile contamination whereas carefully enhanced disinfection did; bleach goes round corners better than UV, apparently. A related (and under-reported) unintended consequence of introduction a “no-touch” room disinfection (NTD) system such as UVC is that cleaners stop cleaning, mistaking UVC for magical cleaning robots! Plus, you could find yourself spending more time screening than cleaning, to the extent that those tasked with monitoring the cleaning process would be better deployed by getting their hands dirty! Dr Donskey covered a number of other important environmental issues: who cleans what (“the nurses thought EVS were doing it; EVS thought the techs were doing it; nobody was doing it”), the need for daily disinfection, pre-emptive and extended isolation, the potential and under-recognized importance of proper daily bathing for CDI patients, and the potential contamination risk from asymptomatic carriers. The final word: “getting doctors to prescribe antibiotics appropriately is like getting EVS to clean properly: an ongoing challenge.” Oh, and he finished on a song.
Jon Otter PhD (who invited him?) – No-touch room disinfection (NTD) systems: when to use them and how to choose between them (Can you ‘C’ the difference?)
You can download my slides from the talk here.
The talk was loosely based around a review paper recently published in JHI. The increased risk from the prior room occupant argues for doing a better job of terminal disinfection. The goal of hospital disinfection is controversial: the ‘Pragmatist’ says a reduction in contamination is good enough, whereas the ‘Prior room occupantist’ says elimination of pathogens is required. I presented some data suggesting that transmission risk ∝ contamination level; ergo reduction in transmission ∝ decontamination level? The NTD scene is a four-horse race currently, with hydrogen peroxide vapour (HPV), aerosolized hydrogen peroxide (AHP), ultraviolet C (UVC), and the relatively new kid on the block, pulsed xenon UV (PX-UV). Each system has its pros and cons so which is best? My view is that will depend on the scenario: if you have a carbapenem-resistant Acinetobacter baumannii in your ICU, then the ‘belt and braces’ approach of HPV is warranted. However, if you have MRSA colonization on a medical ward, a ‘quick and easy’ UV treatment may the only feasible option.
To try to keep everybody awake after lunch, I polled the audience on a few questions (Figure). I was not surprised that most people had not used an NTD system. However, I was surprised that so few people selected UV in the scenarios!
Figure. Question 1: Should all acute hospitals be using a ‘no-touch’ automated room disinfection (NTD) system for terminal disinfection of some patient rooms? Question 2: Has your hospital has used the following NTD systems? Scenario 1: A patient with carbapenem-resistant A. baumannii is discharged from the ICU. Scenario 2: A patient with MRSA colonization is discharged a general medical unit. Scenario 3: A patient with recently resolved CDI is discharged from a general medical unit (‘Enhanced’ = enhanced conventional methods).
Mike McCarthy – Sustaining your gains in infection control initiatives
Mike McCarthy rounded off the day with an engaging overview of his experience from a number of industries of how to ‘sustain your gains’. There’s a temptation from administrators to dismantle the team once it has been shown to work; clearly, the results will disappear with the people! Mike gave useful advice on how to embed change in an individual and organization. Do not confuse respect for people with respect for their bad practices. We need to be good coaches of best practice – reinforce proper execution; correct improper execution. The typical number of audits is “once and done”, but this not enough to form good habits. Establishing a new habit takes 60-90 days of work to reach the happy state of ‘unconscious competence’. People like data-led feedback (we’re all nerds at heart), which results in tangible performance management and improvement. So, implement a checklist, audit it, provide positive reinforcement and feedback and your gain will be sustained!
Points for discussion:
- Laboratory diagnostics are only part of the story. We need to focus on making sure only appropriate specimens are tested. Dr Donskey mentioned that a shocking 12% of their stool specimens were not tested due to sample leaking or labeling errors. Unfortunately, the stools most likely to be from CDI are also most likely to be liquid! Conversely, testing formed stools doesn’t do anybody any favours.
- Do we need to focus on asymptomatic toxigenic C. difficile carriers and, if so, how?
- How far can conventional methods go in tacking environmental contamination with C. difficile and is it time to turn to NTD systems, at least some of the time?
- How best to sustain our gains?
Is “community-acquired” CDI real?
A recent high profile US study delved into apparent community-associated CDI cases to evaluate healthcare exposures. The study was large, evaluating almost 1000 cases of community-associated CDI from 8 US states. Only 177 (18%) of the 984 cases had no recent healthcare exposure (Figure 1). Furthermore, healthcare exposure was only evaluated for the 12 weeks prior to the positive specimen, so I would wager that a portion of this 18% acquired their infecting C. difficile in a healthcare facility. 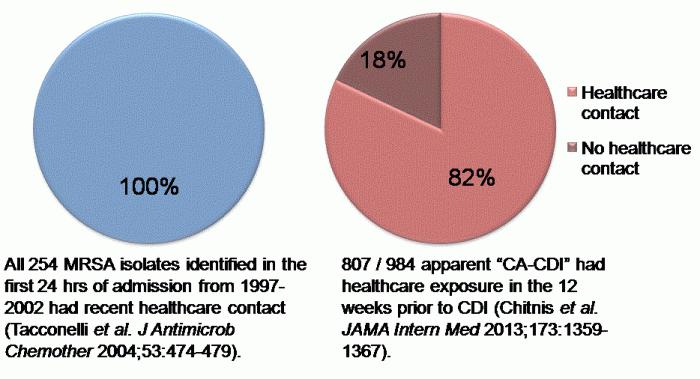 Figure 1. Data demonstrating that most MRSA and CDI presenting on admission to hospital are likely to have been acquired in a healthcare facility.
Figure 1. Data demonstrating that most MRSA and CDI presenting on admission to hospital are likely to have been acquired in a healthcare facility.
So, it seems that the majority of these cases are more likely to be community-onset, healthcare-acquired CDI, rather than community-acquired CDI. I feel like we’ve been here before. In the 1990s before the emergence of distinct strains of CA-MRSA, MRSA presenting at hospital admission was commonly termed ‘community-associated’ or, worse, ‘community-acquired’ when really it was MRSA that had been acquired in hospital during a previous stay (Figure 1). The situation has now changed since distinct MRSA clones have emerged that have the capacity to cause infection outside the healthcare environment.
Turning our attention to the UK, the mandatory report scheme classifies cases of CDI as ‘Trust-apportioned’ if the specimens is collected from patients who have been in hospital for four or more days (Figure 2). It is tempting to speculate that the cases of CDI that are non Trust-apportioned are CA-CDI. However, the definition for ‘Trust-apportioned’ does not account for previous healthcare contact, and the rate of Trust-apportioned and non-Trust-apportioned cases tracks so closely that, once again, these are likely to be healthcare-acquired CDI presenting on admission.
 Figure 2. Number of cases of CDI in England through the mandatory reporting scheme, 2004-2013.
Figure 2. Number of cases of CDI in England through the mandatory reporting scheme, 2004-2013.
The epidemiology of C. difficile is fundamentally different to MRSA, in that healthy neonates typically have a high rate of C. difficile colonization. Thus, there is a ready reservoir for a low rate of genuinely community-acquired CDI. However, it seems to me that most “CA-CDI” reported thus are likely to be acquired in a healthcare facility and I have not seen any data to convince me that community-acquired CDI is increasing.
Article citation: Chitnis et al. Epidemiology of Community-Associated Clostridium difficile Infection, 2009 Through 2011. JAMA Intern Med 2013;173:1359-67.






