 A seminal study recently published in the New England Journal of Medicine evaluated the impact of universal decolonization vs. targeted screening and / or decolonization to prevent ICU infection. The study has the potential to fundamentally change practice in ICUs around the world. However, I have two major problems with the study, namely mupirocin and chlorhexidine.
A seminal study recently published in the New England Journal of Medicine evaluated the impact of universal decolonization vs. targeted screening and / or decolonization to prevent ICU infection. The study has the potential to fundamentally change practice in ICUs around the world. However, I have two major problems with the study, namely mupirocin and chlorhexidine.
Study design and results. The authors describe their design as a ‘pragmatic’ randomized controlled trial, which should serve as a model for other studies. There were several variables that the authors could not change, for example, the fact that some US states mandate universal screening whereas others do not. They were able to cleverly incorporate this into their stratified randomization process so that the integrity of the study was not compromised.
The study was large, with 74,256 patients admitted to 74 ICUs in 43 hospitals randomized to the following groups during the intervention phase:
Group 1: Targeted screening and isolation.
Group 2: Targeted screening, isolation and decolonization (using intranasal mupirocin and chlorhexidine bathing).
Group 3: Universal decolonization (using intranasal mupirocin and chlorhexidine bathing).
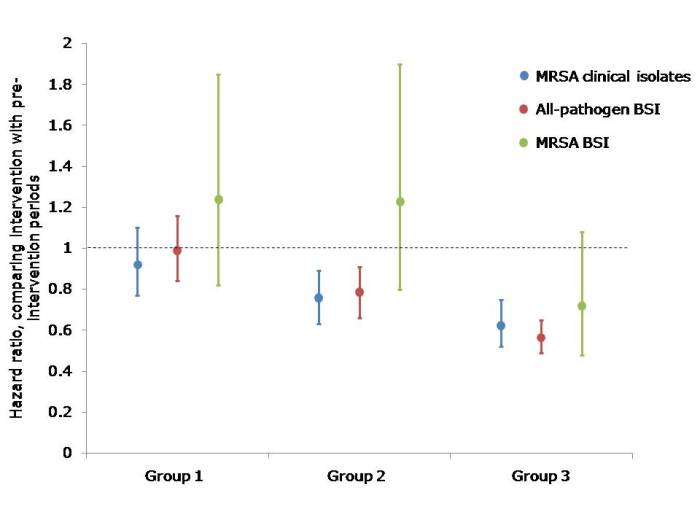
The study compared the hazard ratio for MRSA clinical cultures, all-pathogen bloodstream infections and MRSA bloodstream infections in the 12 month pre-intervention vs. the 18 month intervention period for each of the groups. The main finding was that universal decolonization (Group 3) was associated with significantly lower rates of MRSA clinical cultures and all-pathogen bloodstream infection, and reduced rates of MRSA bloodstream infection (Chart). Target screening and isolation (Group 1) was the least effective strategy and targeted screening, isolation and decolonization (Group 2) had intermediate effectiveness (Chart).
[Chart: hazard ratios and 95% confidence intervals comparing the pre-intervention with the intervention period in each of the three groups. Group 1: Targeted screening and isolation; Group 2: Targeted screening, isolation and decolonization; Group 3: Universal decolonization.]
Major problems: My major issue with the study is the universal (aka indiscriminate) use of mupirocin and, to a lesser extent, chlorhexidine. MRSA are able to acquire low-level resistance to mupirocin through mutations in ileS or high-level resistance through the acquisition of mupA. I can’t help thinking that the authors underestimate the risk of driving mupirocin resistance with their comment in the discussion: ‘Mupirocin resistance has been reported in some studies of MRSA decolonization, but not all such studies.’ It is frankly naive to think that universal use of mupirocin for each ICU admission will not result in the widespread development of mupriocin resistance. Thus, the comment in the accompanying editorial urging caution in implementing the universal use of mupirocin is warranted.
The situation is not as clear-cut with chlorhexidine. Clinically significant reduced susceptibility to chlorhexidine in S. aureus has been hard to define for a number of reasons:
- There is no generally agreed methodology for detecting reduced chlorhexidine susceptibility.
- Population MICs vary considerably making a meaningful breakpoint difficult to determine.
- Higher MICs displayed by some isolates (4–16 mg/L) are still well below the effective chlorhexidine concentration applied to skin (10000–40000 mg/L).
- Although genes encoding membrane efflux pumps have been identified in S. aureus, including qacA, these have an unclear relationship with reduced susceptibility.
- Other mechanisms of reduced susceptibility, such as decreased uptake of chlorhexidine, may confound the apparent effect of efflux pumps.
- Although many studies have reported on qacA carriage or reduced chlorhexidine susceptibility in S. aureus isolates, few have evaluated clinically relevant outcomes.
Notwithstanding these limitations, it is clear that the implementation of chlorhexidine based decolonization drives an increase in the carriage of genes associated with reduced susceptibility to chlorhexidine, and this may be clinically relevant based on studies from London and Geneva. For example, a recent article from my research lab in London found that the implementation of universal chlorhexidine-based decolonization was associated with the selection of qacA linked with a higher chlorhexidine MIC in one dominant endemic MRSA clone (CC22), but not another (CC30). The slower reduction in the CC22 MRSA bloodstream infection rate suggests that carriage of qacA confers a selective advantage.
So, will the introduction of universal decolonization result in resistance to mupirocin and chlorhexidine? The answer for mupirocin is a resounding yes; the answer for chlorhexidine is currently not clear.
Other limitations: It’s always easy to snipe at a high-profile study, but there do seem to be some important limitations. First, nasal screens alone were performed in patients admitted to Groups 1 and 2. The sensitivity of nasal screens alone can be as low as 50%, so the inclusion of other screening sites are preferable to improve sensitivity. The implications of missing many carriers on admission in Groups 1 and 2 are obvious.
Second, although the trend was in the same direction, there was no significant reduction on MRSA bloodstream infections for patients in Group 3. In fact, the crude rate of MRSA bloodstream infection was rather high in all three groups (0.5 to 0.7 per 1000 patient days in all groups during the baseline and intervention periods). For reference, the rate of MRSA bloodstream infection at Guy’s and St. Thomas’ NHS Foundation Trust in London was lower than this at the peak of the MRSA problems in 2003, approximately 10 times lower by 2008 and has fallen further since then. So, why the relatively high rate of MRSA bloodstream infection across the board in this study?
Third, while randomization is the “gold standard” in terms of study design, by chance, three of four hospitals that performed bone marrow and solid organ transplantation ended up in Group 3, which skewed this population somewhat. The study was large enough to “smooth out” this skew, but small studies that attempt randomization should take note. In a recent study with Johns Hopkins, we only had 6 units to play with so decided not to randomize but instead roughly “matched” the units in the two arms, which may be a better approach in smaller studies.
Fourth, it does not seem that any attempt was made to ensure that patients in Groups 1 and 2 were bathed daily. While bathing with chlorhexidine impregnated cloths has been shown to be superior to bathing with soap and water, it is not possible to determine whether daily bathing or chlorhexidine were most important.
Finally, compliance will never be 100%. Only 81% of patients in Group 3 actually received chlorhexidine and 86% received mupriocin, and only approximately 90% of patients in Group 2 that were due decolonization received chlorhexidine and mupriocin. However, the authors did achieve a very high compliance (in the high 90s) with admission screening in Groups 1 and 2.
Summary: The authors should be complimented for performing such a detailed and useful study. Unfortunately, the protocol was not configured to assess the relative contribution of mupirocin and chlorhexidine to the reductions in infection. Given the likely emergence of mupirocin resistance, future studies should drop the universal use of mupirocin all together and see how far universal chlorhexidine alone can go; recent data suggest it will go a long way. Indeed, the UK has managed to make a dramatic, national reduction in the rate of MRSA bloodstream infection without implementing universal mupirocin.
I have grave concerns about the universal use of mupirocin for all ICU admissions and, perish the thought, for all hospital admissions. To decide on implementing universal mupriocin use with a plan to keep an eye on resistance rates seems remiss (to put it politely) since the likelihood of widespread resistance is so high. I am less concerned about the universal use of chlorhexidine, although reduced susceptibility needs to be monitored carefully.
The accompanying editorial was entitled ‘Screening inpatients for MRSA – the case is closed’. For me, the only closed case emerging from this study is that universal, indiscriminate use of mupriocin is a bad idea. Antibiotics have caused the problem; they are not the solution.
Article citation: Huang et al. Targeted versus universal decolonization to prevent ICU infection. New Engl J Med 2013 May 29. [Epub ahead of print].
Discover more from Reflections on Infection Prevention and Control
Subscribe to get the latest posts sent to your email.
Finally a voice of reason from a basic scientist. It has been proven on repeated occasions that resistance will emerge, and will eclipse the intended purpose of the agent. Examine the core problem – MRSA – methicillin resistance emerged almost coincidentally with the increase use of the agent.
LikeLike
Thanks David. As you can probably tell, I feel strongly about this. We have countless examples of antibiotic overuse and resistance: S. aureus and penicillin then methicillin, Gram-negatives and beta-lactams then extended spectrum beta-lactams and now carbapenems. We need to ring-fence and preserve the activity of the effective agents that we have left.
LikeLike
totally in agreement with the message of prudence given.
1- The use chlorhexidine for everything at any time will decrease biologic diversity and increase the potential risk of antiseptic resistance.
2- Gram negative MDRO is nowadays the most important problem especially in Europe. The impact on Gram negative especially KPC, MDR Acinetobacter baumannii and Pseudomonas is probably less pronouced given the previous results published by Climo et al in NEJM 2012 and given the known less important activity of chlorhexidine on gram negative.
a more targeted approach combined with targeted screening is in my opinion by far less dangerous…
Prof Jean-Francois TImsit MD PhD
University hospital Albert Michallon, University Grenoble 1
Grenoble France
LikeLike
Thank-you for taking the time to comment on the blog Prof Timsit,
You’re right in that the potential for resistance to chlorhexidine is greater among the Gram-negatives due to their extended array of biocide and antimicrobial resistance mechanisms. For example, this disturbing study from 2002 shows that several Gram-negative species with substantially reduced susceptibility to chlorhexidine were isolated from skin treated with chlorhexidine at catheter insertion sites. I had not noted the relatively poor impact of chlorhexidine bathing on Gram-negatives in the Climo NEJM study so thanks for highlighting that.
You make an interesting point about reduced biodiversity. I guess the concern here is a “squeezing the balloon” effect, where preventing infections due to chlorhexidine susceptible bacteria leaves a niche for chlorhexidine tolerant / resistant bacteria. Potentially, out of the frying pan and into the fire!
LikeLike
I had a reservation about this issue some time ago when I heard about that. I am following this issue for quite long time which started first as “Targeted Screening on Admission to ICU”, then advance to “Universal Screening” over the past several years and now we are talking about “Universal Decolonization”. I agree to one part of it that is Universal Decolonization with Chlorhexidine Bath but say NO to Mupirocin use due to potential prospective resistance. Even now most healthcare facilities are using Mupirocin Nasal blindly not aware of its resistance as many do not test this drug in lab for C/S against MRSA. So yes I do agree it might tend towards universal resistance later on.
My point of view is mainly clinical I am not a Microbiologist but I am well aware dealing with these situations for many years and also well aware about its outcome. At present, what my protocol for ICU is to do universal Nasal screening but additional screening sites for patients referred from other healthcare facilities or patients with devices. And the good thing is that have a full occupancy 24-bedded ICU and all are single rooms providing opportunity for all admissions to be placed in single rooms or we may call it contact isolation until screening swabs results are available. Single rooms ICU provides us physical barriers between patients and hence probability of cross-transmission of infection or colonization is less likely. About decade ago I introduced an idea of single rooms ICU because we had difficult times with MRSA in our open-type ICU separated by curtains which was really a mess due to fast turn-out and less time for cleaning and disinfection. Therefore as I said above we do universal screening, all patients placed in contact precaution, upon arrival of C/S we either continue or D/C isolation and use Chlorhexidine bath for infected or colonized patients.
But this is not the end of the road, we have to be vigilant what our Intensivists are prescribing. I did so many projects in ICU with Nursing and treating physicians. Unfortunately many Physicians could not interpret well culture reports and they tend to treat the culture rather the patient. I have seen lots of antibiotic overuse but with their cooperation we did address it well. Since Antibiotic Education and Management is my area of interest mainly clinical I spend most of my time with physicians in ICU discussing MDROs and how to manage or interpret a culture report. You must have seen most physicians treating either contamination or colonization and the unfortunate critically ill patients receives unnecessary multiple antibiotics for prolong duration. I have done so much clean-up operation regarding this issue but with physicians cooperation. I carried out numerous antibiotic education sessions whether on site, centralized or unit based and even during medical council or grand rounds with them. As a result our Antibiotic Resistance is declining and antibiotics effectiveness is really improving over the years and with good patient outcomes. And all this evident based on my Antibiograms for ICU which shows gradual decrease in resistance in various types of MDROs. But again I would say I say No to Mupirocin Nasal unless targeted and with clinical indication.
LikeLike
Thanks Fida for your detailed response. Interesting that you mention your transition from an open-plan ICU to 100% single rooms. You may be interested in this review of a study evaluating the impact of ‘privatizing’ an ICU. It shows that the move from an open-plan unit to 100% single rooms significantly reduced the transmission of microbes associated with exogenous spread but made no impact on the transmission of microbes associated with endogenous spread.
It sounds like you’ve been doing some sterling work in restricting the unnecessary use of antibiotics in your own facility, with positive results. I hope that others take heed of this advice and experience.
Finally, at Guy’s and St. Thomas’ NHS Foundation Trust in London where I work part-time, we perform universal screening for MRSA and universal application of chlorhexidine. This allows targeted use of mupirocin for MRSA carriers and realizes the benefits of reducing the microbial load on all patients through the universal use of chlorhexidine. There have been some problems with reduced susceptibility to chlorhexidine associated with an outbreak of one particular clone (TW) and increased carriage of qacA and reduced chlorhexidine susceptibility in a common epidemic clone (EMRSA-15). However, both of these clones have been controlled successfully on our ICU to the extent that MRSA infections and especially bloodstream infections are now rare.
LikeLike