Since writing this 2015 review on gaps and controversies in the guidelines for the prevention and control of CPE (and other MDR-GNR) I’ve tried to keep it fairly up to date. So, here’s the latest iteration, including the 2015 CDC guidelines.
carbapenem
21 is the magic number (for defining CPE person-to-person transmission using WGS)
A fascinating study from a European research group has unravelled the molecular epidemiology of a large European collection of carbapenem-resistant Klebsiella pneumoniae clinical isolates. Most carbapenem resistance was due to an acquired carbapenemases, transmission clusters were common within and between hospitals, carbapenemase-producing isolates are more likely to spread in hospitals, and 21 SNPs is the magic number for defining CPE person-to-person transmission using WGS.
Rampant carbapenem-resistant Klebsiella BSIs: a window to a post-antibiotic apocalypse?
People have been talking in apocalyptic terms for years – probably decades – about the threat of AMR. But has this really materialised? MRSA BSIs are now rare in the UK, and C. difficile infections are rarer than they once were. But things are looking considerably gloomier in other parts of the world. For example, a frankly shocking study from a Greek ICU gives us a view of what a post-antibiotic apocalypse may look like…
The days of serial admission screening for CPE are numbered
The current national guidelines for CPE in England recommend three serial admission screens each separated by 48 hours to confirm a negative carrier status combined with pre-emptive isolation. Even leaving aside the infeasibility of pre-emptive isolation, this approach introduces a host of operational challenges. In a study just published in JHI, we find report that serial admission screens do not improving the detection of CPE. However, there was a striking apparent increase in the rate of carriage of other resistant Gram-negative bacteria in the early days of hospital admission, suggesting either an unmasking of pre-existing carriage or acquisition of resistant Gram-negative gut flora.
CPE has landed in Manchester…officially
A really important point prevalence survey of CPE carriage in inpatients in a hospital in Manchester has just been published in the Journal of Hospital Infection. Important because of the high rate of carriage (11% of 662 patients included). Important because for every 1.6 known cases of CPE, there was an undetected case lurking, despite an extensive screening programme. Important because the lack of significant risk factors associated with CPE carriage suggests that it is distributed homogeneously, endemic in the population. And important because this is the first citable publication suggesting that Manchester has a problem with CPE, despite us having known about it for years via professional networks.
Preventing carbapenem-resistant Salmonella or E.coli in foods
We welcome another guest post from Prof Peter Collignon (bio below) on the risk of antibiotic resistance in the food chain…
Codex has recently announced they are doing more work on antimicrobial resistance. This is an opportunity get Codex to have a couple of standards to better protect public health from one of the worst types of antibiotic resistance that might develop in the next few years or decade. Specifically to help prevent carbapenem-resistant bacteria transferring to people via the food chain including via imported foods into countries (some useful background on this issue from European Food Safety Authority (EFSA) here).
Transmissible colistin resistance – made in China
The emergence of CPE (and carbapenem-resistance in other Gram-negative bacteria) has forced us to reach to the dusty old antibiotic shelf to revive the clinical use of older agents with activity against Gram-negative bacteria, principally colistin. Colistin isn’t perfect by any means – it has poor tissue penetration compared with the carbapenems, and is associated with nephrotoxicity (although the high levels of nephrotoxicity in the older medical literature has not been reported due to better management of the drug). Furthermore, resistance has already been reported. To date – this has been mutational resistance, which does not have the capacity to spread horizontally. It was only a matter of time before a colistin resistance gene mobilised.
What drives carbapenem resistance? Carbapenem use, stupid!
An outstanding study from colleagues at Imperial shows that if you use a bucket-load of meropenem (a carbapenem antibiotic) one year, you see an uptick in carbapenem-resistant Enterobacteriaceae (CRE) the next. The figure below plots meropenem use in the previous year with the incidence rate of OXA-48 producing K. pneumoniae CRE.
Figure: The use of meropenem in the previous year plotted against the incidence rate of OXA-48 producing K. pneumoniae
CRE: coming to a hospital near you
I thought for quite some time about whether the title to this post ought to be a statement or a question. I decided on a statement: pretty much wherever you are in the world, I am certain that CRE is now one (hospital) degree of separation from you.
I gave this talk yesterday at the imaginatively named “Darling Bugs of May” IPS conference, and you can download my slides here. I’ve given similar talks before, but the whole thing took on greater significance now I have had some first hand experience of making decisions around the management of CRE patients.
Do you know your CRE from your CRAB?
I gave a talk today at a meeting on combating carbapenem-resistant organisms. My angle was to clearly differentiate the epidemiology of the Enterobacteriaceae (i.e. CRE) from the non-fermenters (most importantly carbapenem-resistant A. baumannii – CRAB), and you can download my slides here.
I’ve blogged before about how confusing the terminology surrounding multidrug-resistant Gram-negative rods has become. Non-expert healthcare workers have little chance in distinguishing CRE from CPE from CRO from CPO. So we need to help them by developing some clear terminology, given the gulf in epidemiology between CRE and CRAB (see below).
CRE and CRAB are like apples and pears: they share some basic microbiology but that’s about where the comparison ends!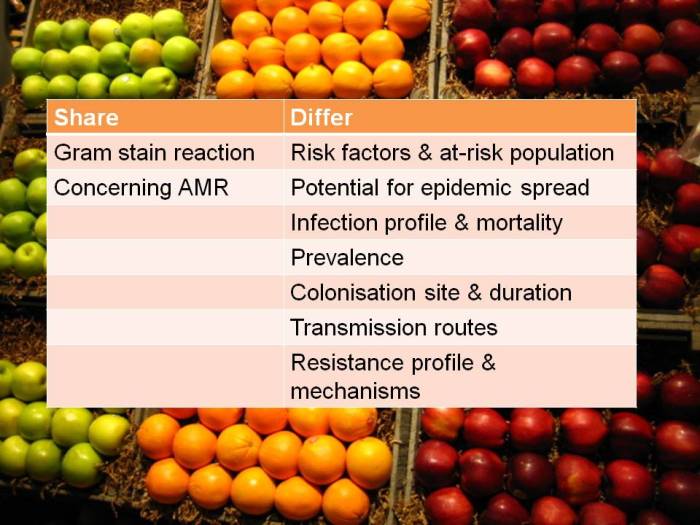
So, I think we should talk in terms of CRE (and CPE for confirmed carbapenemase carriers), and CRNF (or CRAB for A. baumannii and CRPA for P. aeruginosa). I don’t think that CRO is a useful term – in fact, I find it rather confusing. Carbapenem resistance in Enterobacteriaceae (CRE) and A. baumannii (CRAB) are both emerging problems, but they are not the same problem.



