JHI have published a Special Edition featuring write-up from the 2014 HIS Conference (you can see my reflections from HIS here).
- The Special Edition kicks off with a thoughtful editorial by the guest editor Dr Mark Walker, in praise of enthusiasts and in defence of conferences.
- Dr Seto’s Lowbury lecture exploring facts and myths around airborne transmission.
- My overview of trends in the infection prevention and control literature since the 2012 HIS conference (there’s been one trend that trumps all other trends: Ebola). You can download the slides and see a recording of this talk here. Also, Dr Jim Gray’s take on future trends.
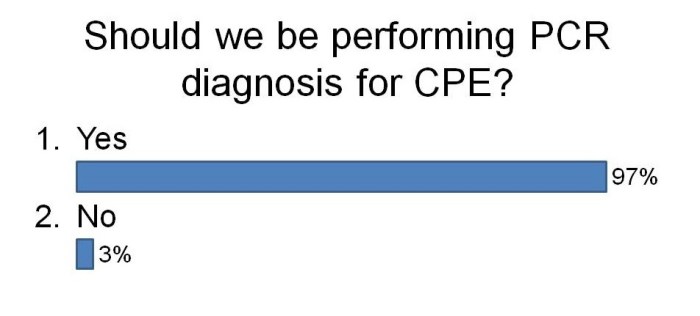
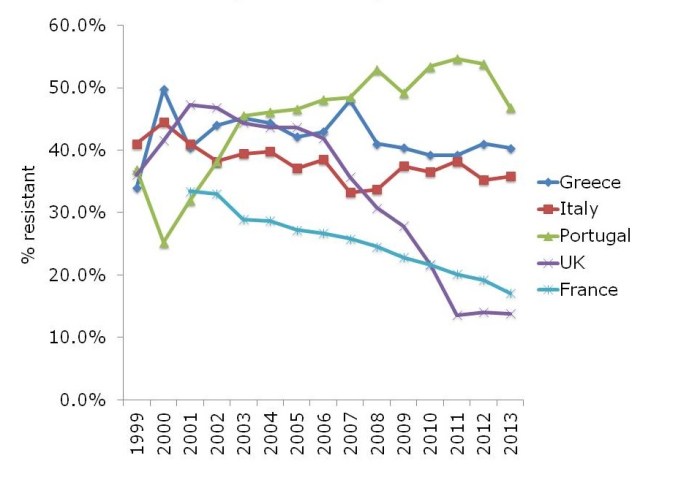
- Updates on multidrug-resistant Gram-negative bacteria, including the importance of globalization, experiences of controlling CRE and Acinetobacter outbreaks, the role of antibiotic prophylaxis in driving the problem.
- Antibiotic stewardship is covered from a couple of angles, including a surgeons’ perspective and long-term care facilities in Ireland.
- Several articles address surveillance, including the role of ‘data warehousing’, integration of electronic systems, and surveillance of ventriculitis following neurosurgery.
- Not much on the role of the environment, although Dr Frederic Barbut provides an overview of methods to eradicate (or not!) C. difficile from the hospital environment.
- State-of-the-art updates on norovirus, Candida, influenza (specifically the role of point-of-care testing), P. aeruginosa, cystic fibrosis, and HCAI in neonates.
- Management and organisational factors are reviewed, and other articles address assessment and public-reporting, root-cause analysis, human factors, training, and current knowledge.
- Finally, and importantly, the impact of the recession on HCAI is reviewed. The recession has hit hard in Ireland, where this article was penned from, but the article at least offers some useful pragmatic coping strategies!
I’m all for special editions, and I think that JHI should do more of them. I know that compiling this Special Edition has been a considerable undertaking for the Journal, but well worth the effort: it’s a very useful read. Particular thanks to Dr Mark Walker who was the editor for this Special Edition, and to Dr Jenny Child who initiated it.