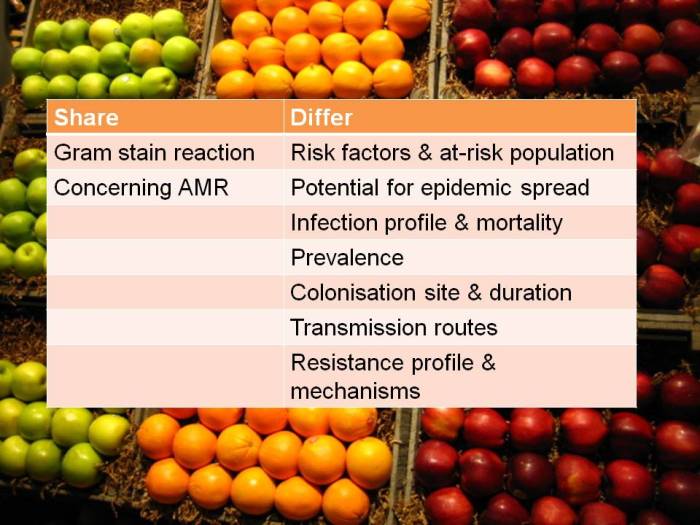
Well, I’m two days in to my first ECCMID meeting, and what can I say, it’s huge. There also seems to be a bit of IPC creeping in, however one thing has struck me. There has been debate as to whether some interventions, for example chlorhexidine washing, is effective but in reality it seems to be common with many interventions in IPC that the real issue isn’t the intervention, it is getting people to reliably implement it that is the real issue.It is the same for pre-op prophylaxis, warming etc., etc., etc. We give feedback on SSI rates to surgeons, but what about the others who have an influence on the bundle that should be implemented. Not seen anyone look at performance of individual anaesthetists or other theatre practitioners in implementing antibiotic prophylaxis, warming, supplemented O2 and glucose control yet they are the gatekeepers of these interventions.
Whether something works in a lab or is theoretically possible seems to me to be a bit academic if no-one will do it. I saw a study today presented by Stephan Harbarth, who was defending screening and decolonisation (I prefer suppression) for Staph infection prevention and the compliance was <50% although there seemed to be an effect. Andreas Voss countered that if we cannot implement an intervention we should not be putting it in place, a fair point. In my humble opinion we really need to undertake some good qualitative studies that look at why interventions that may be (and sometimes that absolutely are) effective are not implemented despite the evidence. Is it that we are unable to personalise the outcome (for staff) or that (in the case of patients) that the perception of risk to the self is low, despite the evidence, as in smoking and alcohol intake. Perhaps we should have to describe how to implement reliably as part of the research and development process for the intervention. Otherwise are we just producing yet another publication or free paper that will not reliably and consistently be implemented and that will never really see the light of day?