I had the opportunity to ask the audience how they were detecting CRE in their diagnostic clinical labs during a talk last week. It was an audience of around 50 laboratory and clinical folk, mainly from the UK but a few from continental Europe. And here’s what I found:
I was a little surprised that more labs have switched to using chromogeneic agar plates than use non-chorogeneic agar plates. In the case of our lab in London, we are currently using non-chromogenic media for clinical samples, but in the process of evaluating chromogenic media. Although the purchase costs of chromogenic media are higher, they are more sensitive and substantially reduce the amount of time required to confirm a negative or positive culture, so I suspect they actually work out cheaper when you factor in labour costs.
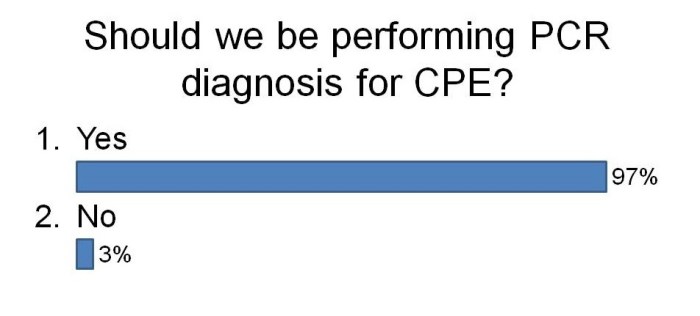
I was not surprised that so few labs are using PCR. The costs are considerably higher but turnaround time is faster and they are more sensitive. There are now a number of PCRs on the market for the detect of CRE direct from rectal swabs (e.g. Checkpoints and Cepheid). We are currently in the process of evaluating the Checkpoints assay and after sharing our preliminary data, this was the feeling in the room about using PCR to detect CRE:
I think I’ll leave it there for now…