This study has just been published in the Journal of Hopsital Infection, showing that the introduction of hydrogen peroxide vapour (HPV) for the terminal disinfection of rooms vacated by patients with CDI was assocaited with a significant reduction in the rate of CDI, from 1.0 to 0.4 cases per 1000 patient days.
HPV
What are we doing to improve hospital room cleaning and disinfection?
I gave a webinar last week for 3M (you can download my slides here) on “Your hospital room can make you sick: How improved cleaning and disinfection can help”. I asked the audience what they were doing to improve cleaning and disinfection, and thought I would share the findings. I don’t know the exact size of the audience (but it’s usually a couple of hundred mainly US based IPC folks), and the audience were allowed to choose any answers that applied to them for the second two questions.
The room lottery: why your hospital room can make you sick
In this era of increasing patient choice, let’s imagine you were offered the choice between two identical looking hospital rooms. Your chances of picking up a multidrug-resistant organism (MDRO) are approximately doubled if you choose the wrong room. But you have no way of knowing which room is safest.
So what explains this lottery? The key information you have not been told is the MDRO status of the previous room occupants. One of the rooms was previously occupied by a patient with C. difficile, and if you choose this room, your risk of developing C. difficile infection doubles. And it’s not just C. difficile – this same association has been demonstrated for MRSA, VRE, Acinetobacter baumannii and Pseudomonas aeruginosa. Underpinning this association is the uncomfortable fact that cleaning and disinfection applied at the time of patient discharge is simply not good enough to protect the incoming patient.
Over the top and into the trenches
 After 12 years working for Bioquell (and part-time at Guy’s and St. Thomas’ / King’s College London since commencing my PhD in 2005), it’s time for me to move onto pastures new. Next week I’ll start working in a leadership role in Infection Prevention and Control at Imperial College NHS Trust in London. It’s a very exciting move for me and I can’t wait to get going. I thought that now would be a good time to reflect on the water under the bridge of the last decade or so (and I hope you’ll forgive my self-indulgence).
After 12 years working for Bioquell (and part-time at Guy’s and St. Thomas’ / King’s College London since commencing my PhD in 2005), it’s time for me to move onto pastures new. Next week I’ll start working in a leadership role in Infection Prevention and Control at Imperial College NHS Trust in London. It’s a very exciting move for me and I can’t wait to get going. I thought that now would be a good time to reflect on the water under the bridge of the last decade or so (and I hope you’ll forgive my self-indulgence).
Over the last decade, the rate of MRSA and C. difficile infection (CDI) in the UK have fallen dramatically.1,2 At the peak of the MRSA epidemic in the early 2000s, there were more than 2000 MRSA bloodstream infections per quarter in England; now there are 10-fold less.2 It’s not certain how this has been achieved, but a combination of factors, including increased governmental focus, are likely responsible. Whilst MRSA is now rare in the UK this is not the case in other European countries and in many other parts of the world, where MRSA remains common.3
In recent years, a new and more troublesome bacterial threat has emerged: carbapenem-resistant Enterobacteriaceae (CRE).4 CRE present the “triple threat” of high levels of antibiotic resistance (including pan-drug resistant strains against which no antibiotics are left), severe clinical consequences (around half of patients with a CRE bloodstream infection will die), and the potential for rapid regional and national spread (illustrated by national outbreaks in Italy, Greece and Israel).4-6 CRE have been described as “nightmare bacteria” by the US CDC and have prompted unprecedented action from CDC, Public Health England (PHE) and other public health agencies, including a national Patient Safety Alert and a letter to all hospital Chief Executives in the UK to ensure that new CRE guidelines are implemented.7,8
Ebola: infection prevention and control considerations
I gave a webinar yesterday on some of the infection prevention and control considerations related to Ebola. You can view the recording and download the slides here.
Whilst preparing the webinar, it occurred to me that the real game changer in the outbreak that made the world take note was the three transmissions of Ebola in developed healthcare systems outside of West Africa. One occurred in Madrid, Spain in early October, and a further two occurred in Dallas, Texas, a few weeks later. Before these in-hospital transmissions, there was a general feeling that developed healthcare systems could handle Ebola safely. Clearly, that was not the case!
Furthermore, the ratio of secondary transmissions for dealing with Ebola cases in developed healthcare systems isn’t great: of the 13 cases that have been cared for outside of West Africa, three secondary transmissions have occurred.
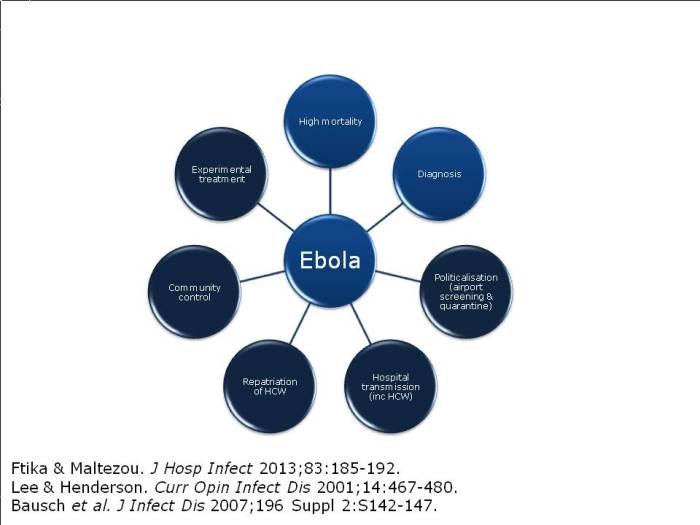
The outbreak has thrown up some new challenges, outlined below.
Figure: the emerging challenges of the Ebola outbreak (the dark shaded circles indicate the new and emerging challenges).
Many of us now find ourselves scrambling to develop Ebola preparedness protocols. These must start at the hospital door, with carefully considered risk assessments for patients presenting with Ebola-like symptoms. We can’t afford to get our full PPE kits out for every patient who presents with a fever, so what should be the trigger for a suspected case? (PHE and CDC have published useful algorithms to help with this, but it’s not straightforward.)
One area of controversy is the appropriate protocols for terminal decontamination following a case of Ebola. Clearly, the most important risk in terms of transmission is direct contact with blood or body fluids from infected patients. However, despite being an enveloped virus, Ebola can surface on dry surfaces for days to weeks under some conditions in laboratory studies. Furthermore, transmission has been associated with indirect contact with contaminated environments. For example, in a recent report from the field, inadequate use of PPE for dealing with surfaces that were grossly contaminated with body fluids from confirmed cases was identified as one of the risk for acquisition. So, we need to make sure that contaminated surfaces are dealt with appropriately, and most hospitals that have dealt with cases outside of West Africa have used hydrogen peroxide vapour for terminal decontamination.
There is a suggestion today that the epi curve may be peaking in Liberia, which is the epicenter of the outbreak in West Africa. Even if that is the case, we can still expect to see more repatriations to developed healthcare systems and perhaps more cases showing up at our hospitals. So, we need to make sure we do everything in our power to prevent secondary in-hospital transmissions.
Is it time to turn to ‘no-touch’ automated room disinfection?
I gave a webinar for 3M yesterday entitled ‘Is it time to turn to ‘no-touch’ automated room disinfection (NTD)?’ It was based broadly on a recent Journal of Hospital Infection review article, and you can access the slides here.
The webinar covered:
- The key data supporting the need for improved hospital disinfection, particularly ‘terminal disinfection’ when patients are discharged.
- The strengths and limitations of conventional disinfection methods, particularly in terms of reliance on the operator to ensure adequate formulation, distribution and contact time of the active agent.
- The potential benefits of introducing automation into the room disinfection process.
- Coverage of the advantages and disadvantages of the various “no-touch” automated room disinfection systems currently available.
- Scenarios in which NTD systems may be warranted.
To summarize the rationale for using an NTD system: enhanced conventional methods are able to eliminate pathogens from surfaces, but the inherent reliance on a human operator to ensure adequate formulation, distribution and contact time of the active agent introduces variability into the process. NTD systems remove or reduce reliance on the operator for delivering hospital disinfection. However, they do not obviate the need for cleaning, so they are designed to augment rather than replace conventional methods.
So when to consider an NTD system? The flow chart below (Figure 1) shows a decision tree for which cleaning and disinfection approach to take. Given their practical limitations, NTD systems are best suited to disinfection of a room after a patient colonized or infected with a pathogen has been discharged to protect the incoming patient from acquiring the pathogen left behind by the prior room occupant. A recent study of a hydrogen peroxide vapor (HPV) NTD system shows that patients admitted to rooms disinfected using HPV were 64% less likely to acquire any multidrug-resistant organism (MDRO) than patients admitted to rooms disinfected using standard methods when the prior room occupant had an MDRO. Figure 1. A disinfection decision diagram for when to consider an NTD system. a) Key pathogens associated with contamination of the environment include C. difficile, VRE, MRSA, A. baumannii, P. aeruginosa and norovirus. b) All NTD systems are applied after a cleaning step to ensure that surfaces are free from visible contamination, which is unacceptable to subsequent patients and will reduce the efficacy of the NTD disinfection. c) There is limited equivocal evidence that enhanced cleaning / disinfection in a low-risk general ward setting can reduce the spread of pathogens.
Figure 1. A disinfection decision diagram for when to consider an NTD system. a) Key pathogens associated with contamination of the environment include C. difficile, VRE, MRSA, A. baumannii, P. aeruginosa and norovirus. b) All NTD systems are applied after a cleaning step to ensure that surfaces are free from visible contamination, which is unacceptable to subsequent patients and will reduce the efficacy of the NTD disinfection. c) There is limited equivocal evidence that enhanced cleaning / disinfection in a low-risk general ward setting can reduce the spread of pathogens.
Ok, so you’ve decided that you want to use an NTD system. Which one to choose? Every conference I go too seems to have more and more NTD systems on show, all with bold and often conflicting claims. There are essentially four classes of NTD system that are commonly used in hospitals:
- Hydrogen peroxide vapor (HPV)
- Aerosolized hydrogen peroxide (aHP)
- Ultraviolet C (UVC)
- Pulsed-xenon UV (PX-UV)
I asked the audience which, if any, NTD system had been used in their hospital (Figure 2). 90% of the predominantly US based audience had not used an NTD system at all, which was a surprise. In the hospitals that had used an NTD system, there was a fairly even split between HPV and the UV systems. Figure 2. Has your hospital used an NTD system and if so, which one?
Figure 2. Has your hospital used an NTD system and if so, which one?
Each of these systems have advantages and disadvantages, which I have tried to summarize in the following table by ranking the systems in the key categories. The hydrogen peroxide systems tend to have higher efficacy and better distribution than the UV systems. But the UV systems are faster and easier to use. Thus, there is a trade-off between efficacy / distribution and cycle time / ease of use when deciding which NTD system would be more appropriate. Table: Comparing the key features of the four commonly used NTD systems.
Table: Comparing the key features of the four commonly used NTD systems.
In order to illustrate the challenges in choosing a) whether to use and NTD system and b) which to use, I presented the audience with three scenarios. In scenario 1, below, I was expecting most people to select ‘conventional methods’ or one of the UV systems, which have both been shown to reduce the burden of contamination without reliably eliminating pathogens. The sheer number of patients with MRSA colonization transferred or discharged from general medical wards means that the additional time for HPV may not be warranted. Scenario 1. What do you do when a patient who was colonized with MRSA has been discharged from a room on a general medical ward?
Scenario 1. What do you do when a patient who was colonized with MRSA has been discharged from a room on a general medical ward?
Scenario 2 is an occasion where you want to be sure that residual contamination has been dealt with so that the incoming susceptible ICU patient will not acquire the virtually untreatable carbapenem-resistant A. baumannii. Therefore, HPV, which is associated with the elimination of pathogens from surfaces, is a rational choice.  Scenario 2: What do you do when a patient who had an infection with carbapenem-resistant A. baumannii has been discharged from an ICU room?
Scenario 2: What do you do when a patient who had an infection with carbapenem-resistant A. baumannii has been discharged from an ICU room?
Scenario 3 is more tricky. While the likelihood of C. difficile spore contamination argues for the higher efficacy of the hydrogen peroxide systems, the number of transfers or discharges of patients with C. difficile on a surgical unit may be high, which argues for the lesser efficacy but faster cycles from the UV systems. The majority of the audience selected HPV in this scenario, considering that the combined risk of the pathogen and specialty required the elimination of C. difficile spores from the room prior to the admission of the next patient.  Scenario 3: What would you do when a patient who had C. difficile infection has been discharged from a room on a surgical unit?
Scenario 3: What would you do when a patient who had C. difficile infection has been discharged from a room on a surgical unit?
To summarize, the use of an NTD system to augment terminal disinfection is warranted in some circumstances. The choice of NTD system will depend on a number of factors, including efficacy, distribution, ease of use, cycle time and cost. The features of the various NTD systems make them best suited to different applications, dictated by the clinical setting and the environmental-pathogenic characteristic of the target pathogen. So, is it time to turn to NTD systems? 52% of the audience voted ‘yes’ at the start of the webinar; 74% voted ‘yes’ at the end! Figure 3: Is it time to turn to ‘no-touch’ automated room disinfection? The audience were asked this question at the start and the end of the webinar, indicating a swing towards the affirmative!
Figure 3: Is it time to turn to ‘no-touch’ automated room disinfection? The audience were asked this question at the start and the end of the webinar, indicating a swing towards the affirmative!
How the implementation of hydrogen peroxide vapour (HPV) could save you money
On a recent trip to the US, I asked five or six hospitals what their policy was for dealing with packaged medical supplies (syringes, dressings etc) from the rooms of patients on precautions for MDROs. The response was startling: every hospital had a different policy. The policies ranged from 100% disposal of supplies for every discharge through a “toss heavy toss light” approach depending on the perceived risk of the patient to 0% disposal. I scoured international guidelines and, save a few organism-specific guidance documents, there is no direction on this issue in the guidelines.
What actually happens is another issue. Hospitals with a 100% disposal policy knew that staff often could not bring themselves to throw away perfectly good, sometimes fairly valuable items (IV sets and the like) for the small risk that the packaging may be contaminated. Some threw their supplies into “third world bins” to send to underprivileged hospitals, which is great, unless they happen to be contaminated with an MDRO that would survive the journey! Conversely, hospitals with a 0% disposal policy know that sometimes staff threw out the supplies if the patients had a high perceived risk of shedding. As for the “toss heavy toss light” hospitals: who knows what actually happens.

So, is there a real risk associated with contamination of the packaging of these items? A recent study by Johns Hopkins published in the recent ICHE special issue sampled a selection of supplies to quantify the risk, counted the cost of their current policy and found a potential solution in the use of hydrogen peroxide vapour (HPV) for the disinfection of the supplies. The study found that the packaging of 7-9% of supply items was contaminated with MDROs, and that hydrogen peroxide vapor (HPV) was effective for the disinfection of the supply packaging. The cost of supplies discarded from six ICUs amounted to almost $400,000, not including the costs associated with waste disposal. Hence, the practice of disinfecting the packaging of supplies using HPV would generate substantial cost savings.
Improved terminal disinfection using hydrogen peroxide vapour (HPV) or bleach to control C. diff
Mitigating the increased risk from the prior room occupant through HPV room disinfection
The role of surface contamination is increasingly recognised in the transmission of certain nosocomial pathogens1. The most compelling evidence comes from the finding that admission to a room previously occupied by a patient infected or colonised with some multidrug-resistant organisms (MDROs) increases the risk of acquiring that MDRO for the subsequent room occupant by a factor of two or more1-3. Conventional cleaning and disinfection does not reliably remove all environmental MDROs4,5. Hence, it seems that inadequate terminal disinfection of hospital rooms explains the association with the increased risk of acquisition from the prior room occupant. It follows, then, that improvements in terminal disinfection should reduce the levels of residual contamination and the transmission of pathogens through this route. A recent study from Johns Hopkins Hospital tested this hypothesis through the introduction of hydrogen peroxide vapour (HPV) terminal disinfection of selected patient rooms6.
A 30-month prospective cohort intervention study was performed on 6 high-risk units (5 ICUs). HPV was implemented on 3 of the units following a 12-month pre-intervention phase. Clinical impact was assessed by a cohort study. Each patient admitted to any study unit during both phases was included in one of three cohorts:
- ‘MDRO-standard’ Patients admitted to a room where the prior room occupant had an MDRO and the room was disinfected using standard methods.
- ‘MDRO-HPV’Patients admitted to a room where the prior room occupant had an MDRO and the room was decontaminated using HPV
- ‘No MDRO-standard’ Patients admitted to a room where the prior room occupant was not known to have an MDRO and the room was disinfected using standard methods.
The key finding was that patients admitted to rooms decontaminated using HPV were 64% less likely to acquire any MDRO (incidence rate ratio [IRR] of the MDRO-HPV vs. MDRO-standard cohorts = 0.36, confidence interval CI=0.19-0.70, p<0.001) (see chart below).
Chart showing the MRDO acquisition rate in the three patient cohorts.

‘MDRO-standard’ = Patients admitted to a room where the prior room occupant had an MDRO and the room was disinfected using standard methods. ‘MDRO-HPV’ = Patients admitted to a room where the prior room occupant had an MDRO and the room was decontaminated using HPV.
‘No MDRO-standard’ = Patients admitted to a room where the prior room occupant was not known to have an MDRO and the room was disinfected using standard methods.
The difference between cohorts was adjusted for patient level variables such as length of stay, morbidities and other variables that could explain the difference. This means that the difference between cohorts is attributable to HPV alone. When broken down into individual MDROs, the largest reduction was shown for VRE (a 75% reduction, p<0.0001). HPV provided a protective effect for the other MDROs assessed (C. difficile, MRSA and MDR-Gram-negative rods), but differences for these individual pathogens were not statistically significant.
Surprisingly, even when the prior room occupant was not known to have an MDRO, HPV reduced the risk of acquisition by 51% (comparing the MDRO-HPV with the No MDRO-standard cohort). You’d expect the acquisition rate in the ‘MDRO-HPV’ cohort to match the ‘No MDRO-standard cohort’. In fact, it is lower. This is likely due to survival of contamination from previous occupants, unrecognised colonisation or introduction by healthcare workers.
Environmental impact was assessed by sampling each patient room on all units monthly for the last 3 months of the pre-intervention phase and the first 6 months of the intervention phase. Swabs were collected from all patient rooms, occupied or unoccupied, regardless of patient status. The overall percentage of rooms contaminated with one or more MDRO was reduced significantly when HPV was in operation. Further, rooms contaminated with multiple MDROs, occasions when the MDRO from room differed from the room occupant’s known MDRO and MDROs cultured from empty rooms were less likely when HPV was in operation.These changes are due to improved terminal disinfection using HPV.
Whilst this study is one of the few to evaluate patient outcomes in addition to environmental impact of a no-touch automated room disinfection system7. it does have several limitations, some of which are highlight in an accompanying editorial8. Firstly, the rooms or units were not randomized to an intervention arm, which could have introduced bias. Secondly, whilst the conventional disinfection methods were optimized prior to the introduction of HPV, more could have been done to improve the efficacy of conventional methods which may have over-estimated the impact of HPV. Thirdly, the low prevalence of acquisition (especially for MDROs besides VRE) made detecting changes in incidence difficult. Fourthly, whilst the infection rates in the three cohorts did not change significantly with study phase and the clinical impact on an individual patient level was impressive, overall unit level changes in rates of infection were not reported.
The study has important implications for the proportion of transmission that is likely to involve contaminated surfaces. Extrapolating unadjusted data from the study indicates that 16.7% of the acquisitions that occurred during the study were attributable directly to the prior room occupant. This figure most likely underestimates the total contribution of contaminated surface to nosocomial transmission because contaminated surfaces are likely to be involved indirectly in transmission during the stay of affected patients1.
In summary, HPV disinfection significantly reduced the risk of patients acquiring MDROs from previous room occupants in high-risk settings. Furthermore, HPV also provides a protective effect even when the prior room occupant was not known to be infected or colonised with an MDRO. These clinical findings are supported by environmental data showing that HPV disinfection improves the efficacy of terminal disinfection, thus reducing environmental contamination. Whilst the study is not without its limitations, it takes the question of how to tackle hospital environmental contamination forward a pace or two.
Article citation: Passaretti CL, Otter JA, Reich NG et al. An evaluation of environmental decontamination with hydrogen peroxide vapor for reducing the risk of patient acquisition of multidrug-resistant organisms. Clin Infect Dis 2013; 56: 27-35.
References:
1. Otter JA, Yezli S, French GL. The role played by contaminated surfaces in the transmission of nosocomial pathogens. Infect Control Hosp Epidemiol 2011; 32: 687-699.
2. Drees M, Snydman D, Schmid Cet al. Prior environmental contamination increases the risk of acquisition of vancomycin-resistant enterococci. Clin Infect Dis 2008; 46: 678-685.
3. Huang SS, Datta R, Platt R. Risk of acquiring antibiotic-resistant bacteria from prior room occupants.Arch Intern Med 2006; 166: 1945-1951.
4. Manian FA, Griesenauer S, Senkel Det al. Isolation of Acinetobacter baumannii complex and methicillin-resistant Staphylococcus aureus from hospital rooms following terminal cleaning and disinfection: can we do better? Infect Control Hosp Epidemiol 2011; 32: 667-672.
5. French GL, Otter JA, Shannon KP, Adams NM, Watling D, Parks MJ. Tackling contamination of the hospital environment by methicillin-resistant Staphylococcus aureus (MRSA): a comparison between conventional terminal cleaning and hydrogen peroxide vapour decontamination. J Hosp Infect 2004; 57: 31-37.
6. Passaretti CL, Otter JA, Reich NGet al. An evaluation of environmental decontamination with hydrogen peroxide vapor for reducing the risk of patient acquisition of multidrug-resistant organisms. Clin Infect Dis2013; 56: 27-35.
7. Otter JA, Yezli S, Perl TM, Barbut F, French GL. Is there a role for “no-touch” automated room disinfection systems in infection prevention and control? Submitted. J Hosp Infect 2013; 83: 1-13.
8. McDonald LC, Arduino M. Climbing the evidentiary hierarchy for environmental infection control. Clin Infect Dis 2013; 56: 36-9.
Hydrogen peroxide vapour vs. aerosol
There has been an awful lot of discussion out in the field about various hydrogen peroxide systems used for “no-touch” automated room disinfection. Comparison of different systems through assessment of individual studies is tricky because different methods are used to assess the effectiveness of the products. Thus, the only way to get an accurate comparison of different technologies is through head-to-head comparisons.
A recently published study compared a hydrogen peroixde vapour (HPV) system (Bioquell) with an aerosolised hydrogen peroxide (aHP) system (ASP Glosair). The independent study was performed by researchers at St. Georges’ Hospital Testing was performed in a 50m3 room with a 13m3 anteroom, representing a single occupancy room with bathroom. For both systems it was found that rooms must be sealed to prevent leakage and room re-entry must be led by a hand held sensor to ensure safety. HPV generally achieved a 6-log reduction of spore BIs and in-house prepared test discs inoculated with MRSA, Clostridium difficile and Acineotbacter baumannii, whereas aHP generally achieved a 4-log reduction or less. The aHP system had reduced efficacy against the catalase-positive A. baumannii with a <2-log reductions in the majority of room locations. HPV was able to penetrate soiling more effectively than aHP and uneven distribution of the active agent within the enclosure was evident for aHP but not for HPV.
It is difficult to produce a laboratory challenge that is truly representative of field conditions, but the authors did a thorough job and used several different ways to measure the efficacy of the products, concluding that ‘the HPV system was safer to operate, slightly faster and achieved a greater level of biological inactivation than the aHP system.’
Article citation: Fu TY, Gent P, Kumar V. Efficacy, efficiency and safety aspects of hydrogen peroixde vapour and aerosolized hydrogen peroixde room disinfection systems. J Hosp Infect 2012; 80: 199-205.