We have been posting for a while about the emerging recognition of CPE contamination of drains in clinical settings, which seems to be fueling some CPE transmission. Until now, there’s been plenty of publications identifying the problem, but very few presenting a solution. In fact, attempts to tackle CPE contamination of drains have had moderate impact, at best. A new short study in ICHE illustrates the potential of a foaming hydrogen-peroxide based disinfectant to tackle contamination with resistant Gram-negative bacteria in drains.
hydrogen peroxide
Hydrogen peroxide vs. Clostridium difficile
This study has just been published in the Journal of Hopsital Infection, showing that the introduction of hydrogen peroxide vapour (HPV) for the terminal disinfection of rooms vacated by patients with CDI was assocaited with a significant reduction in the rate of CDI, from 1.0 to 0.4 cases per 1000 patient days.
Diluting the efficacy of hydrogen peroxide room decontamination?
A somewhat perplexing new study has just been published in the Journal of Hospital Infection comparing the effectiveness of two hydrogen peroxide based automated room decontamination systems: a low-concentration (5%) hydrogen peroxide system (Deprox) and a high-concentration (30%) hydrogen peroxide system (Bioquell).
The study evaluated the impact of the two systems each run in 10 single rooms containing seeded metal discs placed in five locations, with a 6-log load of MRSA, K. pneumoniae, and C. difficile spores. The MRSA and K. pneumoniae were either low soiling (0.03% BSA) or heavy soiling (10% BSA), and the C. difficile spores was either low soiling (0.03% BSA) or in body fluid. In addition, surface samples were taken from 22 surfaces in each room before and after decon using contact plates. The bottom line is that both systems achieved a >5-log reduction on all of the discs (including those with heavy soiling), and there were no real differences in the levels of surface contamination remaining. All this understandably moved the authors to conclude that ‘The starting concentration and mode of delivery of hydrogen peroxide may not improve the efficacy of decontamination in practice.’
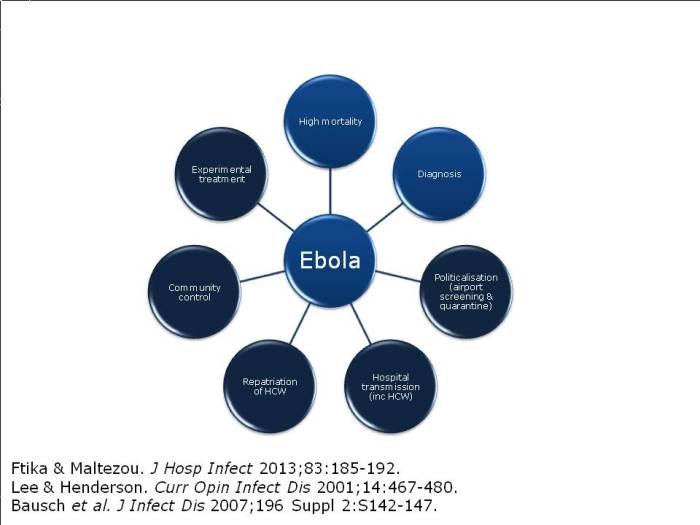
Ebola: infection prevention and control considerations
I gave a webinar yesterday on some of the infection prevention and control considerations related to Ebola. You can view the recording and download the slides here.
Whilst preparing the webinar, it occurred to me that the real game changer in the outbreak that made the world take note was the three transmissions of Ebola in developed healthcare systems outside of West Africa. One occurred in Madrid, Spain in early October, and a further two occurred in Dallas, Texas, a few weeks later. Before these in-hospital transmissions, there was a general feeling that developed healthcare systems could handle Ebola safely. Clearly, that was not the case!
Furthermore, the ratio of secondary transmissions for dealing with Ebola cases in developed healthcare systems isn’t great: of the 13 cases that have been cared for outside of West Africa, three secondary transmissions have occurred.
The outbreak has thrown up some new challenges, outlined below.
Figure: the emerging challenges of the Ebola outbreak (the dark shaded circles indicate the new and emerging challenges).
Many of us now find ourselves scrambling to develop Ebola preparedness protocols. These must start at the hospital door, with carefully considered risk assessments for patients presenting with Ebola-like symptoms. We can’t afford to get our full PPE kits out for every patient who presents with a fever, so what should be the trigger for a suspected case? (PHE and CDC have published useful algorithms to help with this, but it’s not straightforward.)
One area of controversy is the appropriate protocols for terminal decontamination following a case of Ebola. Clearly, the most important risk in terms of transmission is direct contact with blood or body fluids from infected patients. However, despite being an enveloped virus, Ebola can surface on dry surfaces for days to weeks under some conditions in laboratory studies. Furthermore, transmission has been associated with indirect contact with contaminated environments. For example, in a recent report from the field, inadequate use of PPE for dealing with surfaces that were grossly contaminated with body fluids from confirmed cases was identified as one of the risk for acquisition. So, we need to make sure that contaminated surfaces are dealt with appropriately, and most hospitals that have dealt with cases outside of West Africa have used hydrogen peroxide vapour for terminal decontamination.
There is a suggestion today that the epi curve may be peaking in Liberia, which is the epicenter of the outbreak in West Africa. Even if that is the case, we can still expect to see more repatriations to developed healthcare systems and perhaps more cases showing up at our hospitals. So, we need to make sure we do everything in our power to prevent secondary in-hospital transmissions.
Is it time to turn to ‘no-touch’ automated room disinfection?
I gave a webinar for 3M yesterday entitled ‘Is it time to turn to ‘no-touch’ automated room disinfection (NTD)?’ It was based broadly on a recent Journal of Hospital Infection review article, and you can access the slides here.
The webinar covered:
- The key data supporting the need for improved hospital disinfection, particularly ‘terminal disinfection’ when patients are discharged.
- The strengths and limitations of conventional disinfection methods, particularly in terms of reliance on the operator to ensure adequate formulation, distribution and contact time of the active agent.
- The potential benefits of introducing automation into the room disinfection process.
- Coverage of the advantages and disadvantages of the various “no-touch” automated room disinfection systems currently available.
- Scenarios in which NTD systems may be warranted.
To summarize the rationale for using an NTD system: enhanced conventional methods are able to eliminate pathogens from surfaces, but the inherent reliance on a human operator to ensure adequate formulation, distribution and contact time of the active agent introduces variability into the process. NTD systems remove or reduce reliance on the operator for delivering hospital disinfection. However, they do not obviate the need for cleaning, so they are designed to augment rather than replace conventional methods.
So when to consider an NTD system? The flow chart below (Figure 1) shows a decision tree for which cleaning and disinfection approach to take. Given their practical limitations, NTD systems are best suited to disinfection of a room after a patient colonized or infected with a pathogen has been discharged to protect the incoming patient from acquiring the pathogen left behind by the prior room occupant. A recent study of a hydrogen peroxide vapor (HPV) NTD system shows that patients admitted to rooms disinfected using HPV were 64% less likely to acquire any multidrug-resistant organism (MDRO) than patients admitted to rooms disinfected using standard methods when the prior room occupant had an MDRO. Figure 1. A disinfection decision diagram for when to consider an NTD system. a) Key pathogens associated with contamination of the environment include C. difficile, VRE, MRSA, A. baumannii, P. aeruginosa and norovirus. b) All NTD systems are applied after a cleaning step to ensure that surfaces are free from visible contamination, which is unacceptable to subsequent patients and will reduce the efficacy of the NTD disinfection. c) There is limited equivocal evidence that enhanced cleaning / disinfection in a low-risk general ward setting can reduce the spread of pathogens.
Figure 1. A disinfection decision diagram for when to consider an NTD system. a) Key pathogens associated with contamination of the environment include C. difficile, VRE, MRSA, A. baumannii, P. aeruginosa and norovirus. b) All NTD systems are applied after a cleaning step to ensure that surfaces are free from visible contamination, which is unacceptable to subsequent patients and will reduce the efficacy of the NTD disinfection. c) There is limited equivocal evidence that enhanced cleaning / disinfection in a low-risk general ward setting can reduce the spread of pathogens.
Ok, so you’ve decided that you want to use an NTD system. Which one to choose? Every conference I go too seems to have more and more NTD systems on show, all with bold and often conflicting claims. There are essentially four classes of NTD system that are commonly used in hospitals:
- Hydrogen peroxide vapor (HPV)
- Aerosolized hydrogen peroxide (aHP)
- Ultraviolet C (UVC)
- Pulsed-xenon UV (PX-UV)
I asked the audience which, if any, NTD system had been used in their hospital (Figure 2). 90% of the predominantly US based audience had not used an NTD system at all, which was a surprise. In the hospitals that had used an NTD system, there was a fairly even split between HPV and the UV systems. Figure 2. Has your hospital used an NTD system and if so, which one?
Figure 2. Has your hospital used an NTD system and if so, which one?
Each of these systems have advantages and disadvantages, which I have tried to summarize in the following table by ranking the systems in the key categories. The hydrogen peroxide systems tend to have higher efficacy and better distribution than the UV systems. But the UV systems are faster and easier to use. Thus, there is a trade-off between efficacy / distribution and cycle time / ease of use when deciding which NTD system would be more appropriate. Table: Comparing the key features of the four commonly used NTD systems.
Table: Comparing the key features of the four commonly used NTD systems.
In order to illustrate the challenges in choosing a) whether to use and NTD system and b) which to use, I presented the audience with three scenarios. In scenario 1, below, I was expecting most people to select ‘conventional methods’ or one of the UV systems, which have both been shown to reduce the burden of contamination without reliably eliminating pathogens. The sheer number of patients with MRSA colonization transferred or discharged from general medical wards means that the additional time for HPV may not be warranted. Scenario 1. What do you do when a patient who was colonized with MRSA has been discharged from a room on a general medical ward?
Scenario 1. What do you do when a patient who was colonized with MRSA has been discharged from a room on a general medical ward?
Scenario 2 is an occasion where you want to be sure that residual contamination has been dealt with so that the incoming susceptible ICU patient will not acquire the virtually untreatable carbapenem-resistant A. baumannii. Therefore, HPV, which is associated with the elimination of pathogens from surfaces, is a rational choice.  Scenario 2: What do you do when a patient who had an infection with carbapenem-resistant A. baumannii has been discharged from an ICU room?
Scenario 2: What do you do when a patient who had an infection with carbapenem-resistant A. baumannii has been discharged from an ICU room?
Scenario 3 is more tricky. While the likelihood of C. difficile spore contamination argues for the higher efficacy of the hydrogen peroxide systems, the number of transfers or discharges of patients with C. difficile on a surgical unit may be high, which argues for the lesser efficacy but faster cycles from the UV systems. The majority of the audience selected HPV in this scenario, considering that the combined risk of the pathogen and specialty required the elimination of C. difficile spores from the room prior to the admission of the next patient.  Scenario 3: What would you do when a patient who had C. difficile infection has been discharged from a room on a surgical unit?
Scenario 3: What would you do when a patient who had C. difficile infection has been discharged from a room on a surgical unit?
To summarize, the use of an NTD system to augment terminal disinfection is warranted in some circumstances. The choice of NTD system will depend on a number of factors, including efficacy, distribution, ease of use, cycle time and cost. The features of the various NTD systems make them best suited to different applications, dictated by the clinical setting and the environmental-pathogenic characteristic of the target pathogen. So, is it time to turn to NTD systems? 52% of the audience voted ‘yes’ at the start of the webinar; 74% voted ‘yes’ at the end! Figure 3: Is it time to turn to ‘no-touch’ automated room disinfection? The audience were asked this question at the start and the end of the webinar, indicating a swing towards the affirmative!
Figure 3: Is it time to turn to ‘no-touch’ automated room disinfection? The audience were asked this question at the start and the end of the webinar, indicating a swing towards the affirmative!