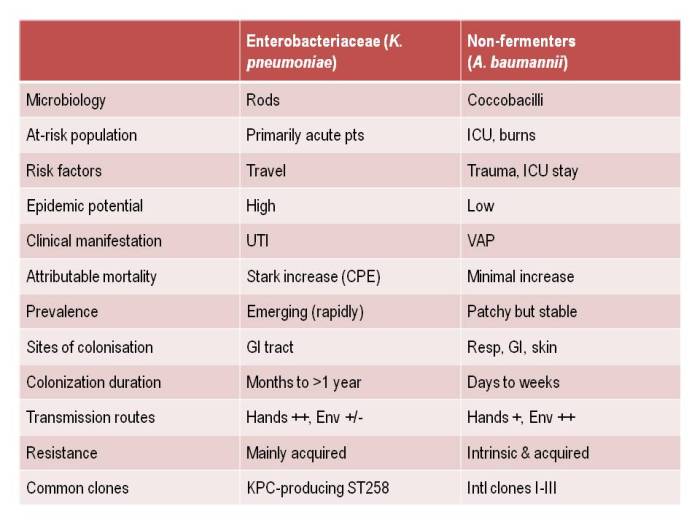
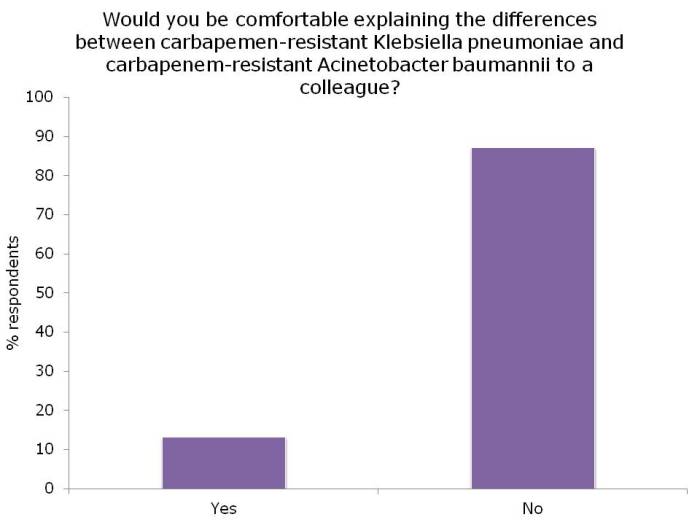
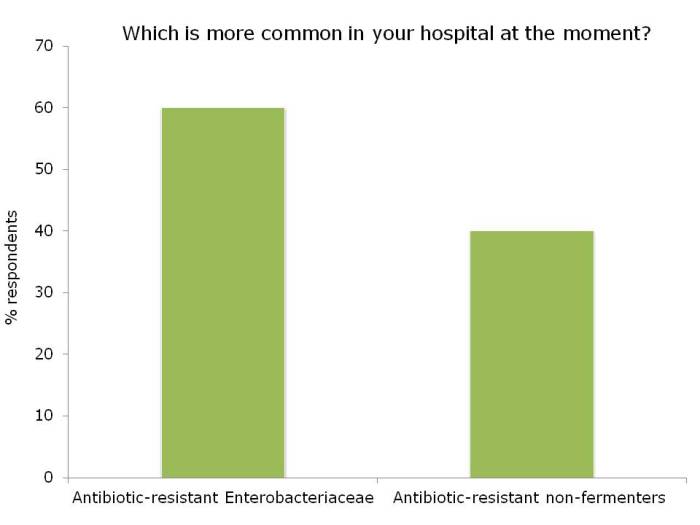
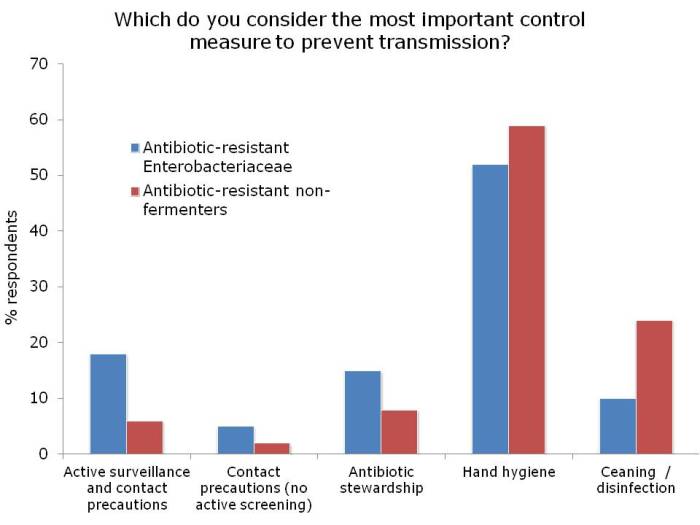
Ok, so the title may be a little premature, since this blog relates to a study with a sample size of exactly one. However, I do think it spells the beginning of the end for Selective Digestive Decontamination (SDD), especially when applied to suppress gut colonization with antibiotic-resistant bacteria.
A number of groups have looked at using SDD to ‘decolonise’ carriers of multidrug-resistant Gram-negative bacteria such as CRE. In one study, 20 CRE colonized patients in each arm given gentamicin + polymyxin (SDD arm) or placebo (Control arm). The results were rather modest (see chart below). Plus, SDD has substantial downsides in terms of the potential for developing further antibiotic resistance, and ‘collateral’ damage to the gut microbiota.
Figure: Modest impact of SDD to ‘decolonise’ the gastrointestinal tract of CRE carriers.
I’ve been waiting for some data on the effectiveness of faecal microbiota transplantation (FMT) to decolonise carriers of antibiotic resistant bacteria for some time. A case report at ID Week related how the ordeal of a 13 year old girl was ended by a faecal microbiota transplantation. After months of persistent colonization and infection, the impact of a single dose of FMT was startling: CRE carriage was eliminated and there was no further bacterial infection.
One of the push-backs against using FMT more regularly is that it’s a crude (in every sense) and labour-intensive procedure compared with an antibiotic capsule. But that was before the invention of ‘crapsules’ (aka oral FMT). Another ID Week abstract reports the successful delivery of oral FMT using crapsules. (And it’s amazing what great dinner party conversation ‘crapsules’ makes. Try it – you’ll see.)
So, I think it’s time for a cluster randomized trial to compare the impact of SDD and FMT; my money is on FMT!
Image: Barbara Krawcowicz.