We are in desperate need of antibiotic-sparing approaches to antibacterial therapy. Antibiotic resistance is increasing, and we are becoming increasingly aware of the impact of antibotics on the microbiota. I blogged a while ago about CRISPR-Cas systems being used to tackle antibiotic-resistant bacteria on surfaces. But the same approach could be applied to treating human infections.
antibiotics
Which is more important? Antibiotic resistance or pyelonephritis?
As we move inexorably towards the end of antibiotics, antibiotic-sparing approaches to the management of infectious diseases become more and more attractive. A study published recently in the BMJ compared the ‘symptomatic’ treatment of uncomplicated UTI in women in the community using ibuprofen with antibiotic treatment using fosfomycin.
Is it true that I have to finish my antibiotic treatment as prescribed?

It is always the same. If I think that I had a reasonable idea and put it on-line, it will only take a day or two, until friends and colleagues let me know, that others had the same idea – just earlier. In this case, John Ferguson (@mdjkf), brought to my attention, that Professor Lyn Gilbert started the same discussion in Australia (Link). As always, I am depressed for about 5 seconds and start to come up with a new, old idea.
Despite the discussion in Down-under, I have promised to post the results of my questionnaire. On Twitter, some were surprised how little evidence exists to support antibiotic prescribing dogmas (Eli Perencevich, @eliowa), whereas others like Christina Vandenbroucke-Grauls (@ChristinaGrauls) believe that we have plenty of evidence from lab experiments and theory to answer the question. Most importantly, “every bit of antibiotic leads to resistance”.
Antibiotic Awareness: background and links
European Survey of Carbapenemase-Producing Enterobacteriaceae (EuSCAPE) project
The EuSCAPE project aimed to improve understanding of the occurrence and spread of CPE. LINK

About European Antibiotic Awareness Day
European Antibiotic Awareness Day is a European health initiative coordinated by ECDC which aims to provide a platform and support for national campaigns on the prudent use of antibiotics. Each year across Europe, the European Antibiotic Awareness Day is marked by national campaigns on the prudent use of antibiotics during the week of 18 November. Prudent use means only using antibiotics when they are needed, with the correct dose, dosage intervals and duration of the course. Follow the European Antibiotic Awareness Day: #EAAD. http://antibiotic.ecdc.europa.eu
About World Antibiotic Awareness Week
The World Health Organization is leading a global campaign for the first World Antibiotic Awareness Week with the slogan “Antibiotics: Handle with Care”. The campaign calls on individuals, governments and health and agriculture professionals to take action to address this urgent health problem. The first World Antibiotic Awareness Week will take place on 16-22 November. Follow the World Antibiotic Awareness Week: #AntibioticResistance. www.who.int/drugresistance.
Continue reading
European Antibiotic Awareness Day 2015: ESPAUR report highlights progress and challenges
For those of you who have had your head in a hole all week, you may not be aware that today is European Antibiotic Awareness Day, which coincides with Global Antibiotic Awareness Week. The antibiotic stewards amongst us (which should really be all of us!) have launched many and varied campaigns to highlight the need to handle antibiotics, our ‘miracle drugs’, with care (see Andreas’ 30-second-antibiotics-myth-buster-survey, for example).
What drives carbapenem resistance? Carbapenem use, stupid!
An outstanding study from colleagues at Imperial shows that if you use a bucket-load of meropenem (a carbapenem antibiotic) one year, you see an uptick in carbapenem-resistant Enterobacteriaceae (CRE) the next. The figure below plots meropenem use in the previous year with the incidence rate of OXA-48 producing K. pneumoniae CRE.
Figure: The use of meropenem in the previous year plotted against the incidence rate of OXA-48 producing K. pneumoniae
Journal Roundup: Ebola, antibiotic abuse, and the usual suspects
The latest edition of Journal Roundup is now available on the Journal of Hospital Infection website, freely accessible here.
Some highlights:
- Ebola science continues to make waves in the big journals. One of the most interesting aspects of the new developments prompted by the Ebola outbreak in West Africa is real advances in vaccine development for Ebola and related viruses.
- We still use a lot more antibiotics than we need to. Why should 50% of inpatients in US hospitals be on antibiotics but only 35% in European hospitals? Is it really that different on either side of the Atlantic?
- Selective digestive decontamination (SDD) results in more pronounced increases in antibiotic resistant bacteria.
- One of the key arguments against contact precautions is an increase in the rate of adverse events due to lower observation. However, a study found that abandoning contact precautions for MRSA and VRE didn’t affect the rate of adverse events. Could it be that the increase in adverse events blamed on contact precautions is explained by underlying risk factors?
- Reference to an outbreak of NDM-producing CRE, prompting calls for sterilization of duodenoscopes (made especially topical by the recent duodenoscope-associated outbreak of CRE in LA).
- Seeding hospital surfaces with Bacillus spores to prevent contamination with multidrug-resistant organisms. That just can’t be a good idea!
- Some brief updates from ID Week in Philadelphia and HIS in Lyon.
Journal Roundup is changing! As a result of feedback received from six months of Journal Roundup, I’ve decided to change the format to write a little more detail on fewer articles. This gives me a good opportunity to accept submissions from others to include in the Roundup. So, Journal Roundup is seeking submissions! If you read an article that you think should be included in the Journal Roundup, please submit a short critique (100 to 300 words) of the article as a comment below to be considered for inclusion in a future edition of the Roundup. I won’t publish the comment on this blog, but your contribution will be acknowledged, of course!
Let’s get those submissions rolling in!
Image: Bacterial spores.
Teixobactin: a “resistance proof” antibiotic? No chance!
It’s not often that I feature a mighty Nature paper on this ‘lil old blog, but this is a big one. A team from Northeastern University in Boston and a small company called NovoBiotic Pharmaceuticals have discovered a truly novel antibiotic, called ‘teixobactin’.
The finding stems from the fact that 99% of microbes in the external environment cannot be cultured in the laboratory. In order to overcome this problem, the authors ingeniously brought the laboratory to the soil by using the iChip (pictured below). The iChip is a way of capturing the growth of a microbe in its natural environment. Curiously, once grown in the iChip, most of the colonies could then be sub-cultured in the laboratory. When using the iChip, around 50% of the microbes in the soil can be cultured (compared with 1% using conventional methods).
The authors then screened extracts from an awful lot of isolates (10,000) for antimicrobial activity, and found one that stood out: ‘teixobactin’. It’s a novel cell wall inhibitor that interrupts peptidoglycan synthesis not by targeting proteins (such as the enzymes targeted by β-lactams), but by targeting lipids.
Teixobactin has impressive activity against a range of Gram-positive pathogens of importance to healthcare including S. aureus, E. faecium / faecalis, various streptococci, M. tuberculosis and, importantly, C. difficile. The authors found that teixobactin had equivalent activity to oxacillin (methicillin) in vitro, and superior activity to vancomycin both in vitro and in an animal model.
However, there are some problems:
- Firstly, and most importantly, there is no activity against Gram-negative bacteria. Since the source microbe, the newly described Elftheria terrae, is a Gram-negative bacterium, this is no surprise, otherwise it would inhibit itself in the soil!
- Secondly, the antibiotic is still a long way from the clinic, and has to undergo a series of rigorous human clinical trials before reaching the pharmacy shelves.
- Thirdly, the authors made the promising discovery that they did not identify reduced susceptibility to teixobactin despite serial passage to sub-inhibitory doses for 27 days. The press have had a field day with this, and are talking in terms of “resistance resistant” antibiotics. But this is too much: the authors parallel the potential for resistance to teixobactin with the potential for resistance to vancomycin – and we are increasingly seeing clinically meaningful reduced susceptibility to vancomycin. There’s a rather obscure and quite frightening study showing that vancomycin resistance could be just around the corner: the study found that S. aureus exposed to sub-lethal doses of chlorhexidine as a surface biofilm became resistant to vancomycin after 48 hours (MIC >128 mg/L). So, bacteria will become resistant to whatever we throw at them, to a lesser or greater degree, given time and sub-lethal exposure.
So, teixobactin represents and exciting and huge leap forward in the process of antibiotic drug discovery – and we can expect more novel antibiotics to follow. However, we’d be foolish to think that resistance to teixobactin will not emerge in time.
Image: NBC news.
Reflections from HIS 2014, Part III: Education, communication, and antibiotic resistance
Welcome to the third and final installment of my reflections from HIS 2014. You can access the ‘box set’ via the list at the start of Part I.
Prof Alison Holmes – Impact of organisations on healthcare-associated infection
Self-professed pragmatist Prof Holmes reminded us that the perceived and actual priorities of society, politicians and healthcare systems mean that it’s not all about infection control. We must harness macro (inter-hospital) and meso (inter-departmental) and micro (inter-team) relationships to successfully control transmission. This requires shared beliefs, reinforcement systems, role models, and the right staff skills. Plus, we need to get HCAI on the metric dashboard of CEOs. Indeed, HCAI outcomes are a sensitive surrogate marker of organisation performance, so this should be attractive to the hospital CEO once understood. We also need to embrace the public to tackle antibiotic resistance. Government messages about reducing antibiotic resistance have helped our day job (and proved popular on Twitter)! Involving patients and the public in our research makes everybody happy; patients and the public like it, and it improves our research (and helps to win grants). We need to embrace ‘mHealth’ in all its forms – games, apps and more – remembering that dinosaurs became extinct. The bottom line? Organisational, structural and managerial issues are crucial for the prevention of healthcare-associated infection (and the Lancet ID agrees).
Prof Herman Goossens – European Antibiotic Awareness Day
Since it was the occasion of European Antibiotic Awareness Day (EAAD) 2014, the talk from the impressive Prof Goossens was well timed! EAAD is a campaign aimed at the public and professionals to highlight the issues around antibiotic use and resistance. Many of the campaign materials are useful, including a toolkit for self-medication without antibiotics and various infographics. Prof Goossens spent some time discussing how to measure the impact of EAAD. A lot of questionnaire type surveys have been performed, and it does seem that EAAD has prompted a swing towards a better understanding of antibiotics, so well done to all involved.
What’s hot and what’s not in infection prevention and control?
Dr Jenny Child (JHI Editor) presented a view of the literature through the eyes of a journal editor! Bad research can do much damage: look no further than the MMR & autism debacle. Worth remembering that indifferent, uncitable papers will not get published; it’s just not in the journal’s interest. Also, clever, ‘pseudo-scientific’ language is a barrier to good science. The bigger the journal, the plainer the language. Finally, whilst JHI has traditionally been a quantitative medicine journal (with p values and 95% confidence intervals!), like it or not, social science is coming!
I gave a talk on ‘What’s trending in the infection prevention and control literature’. You can access the slides and recording on a separate blog, here. Finally, Dr Jim Gray (JHI Deputy Editor) scanned the horizon of the infection control literature, seeing studies with specific interventions and real clinical outcomes (not proxy measures), SSIs, antibiotic resistance (especially CRE), obesity, design & technology, diagnostics and decontamination!
Antibiotic stewardship: persuasion or restriction?
Esmita Charani began by explaining the need to achieve behaviour change, not education in isolation, in order to effectively moderate antibiotic prescribing behaviour. The local prescribing culture is likely to influence prescribing policy more than the national guidelines. Junior doctors often don’t have a clue what to prescribe, so it’s a case of follow-my-leader (i.e. consultant). But targeting hospital consults alone won’t get us out of the mess of antibiotic resistance. We need to engage a wider audience, including the public.
Meanwhile, Prof Inge Gyssens outlined the impact of antimicrobial restriction: contribute to MRSA reductions, prevent the emergence of MDR-GNR, and may help to bring outbreaks under control. The only downside is that switching to another antibiotic may cause more problems than it solves – a ‘squeezing of the balloon’ type effect. In a way, doctors are “addicted” to antibiotics. Put simply, antibiotic stewardship through restriction is a ‘cold turkey’ approach that works.
Although not a formal debate, Esmita Charani and Prof Gyssens did a good job of presenting both viewpoints. I was left concluding that both persuasion and restriction are important but when it comes down to it, restriction is more important than persuasion. Left to their own devices, antibiotic prescribers will sometimes make poor choices; restriction takes away that choice!
Summary
I really enjoyed HIS 2014 – especially the opportunity to contribute to the conference via my talk (on trends in the IPC literature) and poster round. The conclusion of my talk was to look into my crystal ball and highlight what will be trending by the time HIS 2016 comes around:
- I’m pretty certain that Ebola and MERS will not be trending (at least I hope not). However, the scars of Ebola in West Africa will take a generation to heal. There’s a chance that we could be experiencing the next Influenza pandemic, but it’s more likely we’ll be talking pandemic preparedness.
- Whilst I personally favour targeted interventions, I fear there will be a general move towards universal interventions. I also fear that the confusing ‘vertical’ (aka targeted) vs. ‘horizontal’ (aka universal) terminology will be widely adopted, despite the fact that it’s confusing!
- Faecal microbiota transplantation is only going to get bigger. It will be the standard of care for recurrent CDI by the time the next HIS conference comes around – perhaps even via oral ‘crapsules’.
- Whole genome sequencing will not be as trendy as it is right now – it will just be a standard tool that we all use.
- The trend of CRE (and other multidrug-resistant Gram-negatives) is only going to go one way – upwards!
- I’m hoping to see some high-quality studies (ideally cluster RCTs) of environmental interventions with clinical outcomes.
- Finally, as we all deal with increasing cost constraints, studies evaluating the cost-effeteness of infection prevention and control interventions are going to become increasingly important.
A postcard from Portugal: “Some days we don’t have any needles on the ICU”
Most of us know that Portugal is facing a dual threat: high rates of antibiotic-resistant bacteria and financial difficulties. This results in a vicious cycle: there’s no money to address antibiotic resistance, so transmission continues unabated and the antibiotic resistance problem gets worse. You can understand the dilemma from the hospital administrators’ viewpoint: I met an intensivist who confessed that “some days we don’t have any needles”. In this situation, is it better to buy some needles or invest in another infection preventionist?
I recently attended a national infection control meeting in Portugal, where I participated in a forum on “International experiences with HCAI”. You can download my slides here.
MRSA first emerged as a problem in the 1980s in Europe. It became a major problem in many European countries in the 1990s and 2000s so that recent data from ECDC shows high rates of meticillin resistance in S. aureus invasive isolates, especially in some southern European countries; the contrast between the rate of MRSA in the UK and Portugal is stark. In the early 2000s, the rate of MRSA was higher in the UK than in Portugal whereas now, it is much lower in the UK (Figure 1).
Figure 1: Rates of meticillin-resistance in invasive S. aureus in the UK and Portugal. Data from EARS-Net.
Greece, Italy and Portugal are especially affected, with 25 to >50% of invasive S. aureus isolates resistant to methicillin. In the UK, a national strategy has yielded a dramatic reduction in the number of MRSA bloodstream isolates reported to the government in a mandatory reporting scheme (Figure 2).
Figure 2: Dramatic reductions in MRSA bacteraemia in England. But what has made the difference? 
Since the national intervention in England was multifactorial, it is not clear what made the most impact, and it seems likely that more than one intervention contributed to the decline. Interventions included increased attention to intravenous line care, cleaning and disinfection of the environment, improved diagnostics (including the introduction of chromeagar and rapid PCR) and a national hand hygiene campaign. Perhaps the single most important intervention was the introduction of MRSA reduction targets, which were very controversial at the time, but put the issue of MRSA higher on the priority list for the hospital administration.
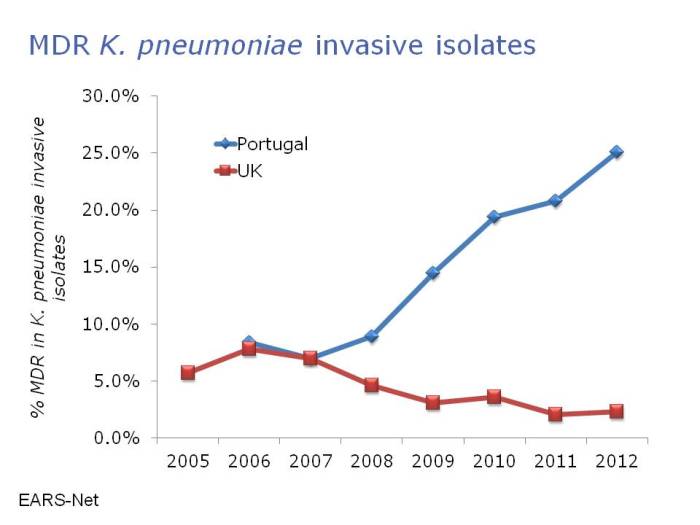
And this issue is not restricted to MRSA. In fact, the threat of the resistant Gram-negatives is even greater than MRSA in many ways. Carbapenem-resistant Enterobacteriaceae are rare currently in Portugal, accounting for 1-5% of invasive K. pneumoniae isolates. However, you get the feeling that it’s only a matter of time: carbapenem-resistant Acinetobacter baumannii are now endemic on many Portugese ICUs, and carbapenem use in Portugal is some of the highest in Europe, with >45% of patients on an antibiotic and >5% of patients on a carbapenem according to the ECDC point prevalence survey. Indeed, there has been a disturbing increase in multidrug-resistant K. pneumoniae in Portugal in recent years (Figure 3).
Figure 3: Disturbing emergence of multidrug-resistant Klebsiella pneumoniae in Portugal. Data from EARS-Net.
The reason for these differences between the UK and Portugal is not clear, but may include infection control staffing, antibiotic usage and lower prioritisation by hospitals. Some progress is being made in Portugal with the recent launch of a national strategy to control healthcare-associated infection. However, the financial climate and somewhat fragmented healthcare system (compared with the NHS) will make implementation challenging. But at least it’s a start.
Image: Portugal stamp.