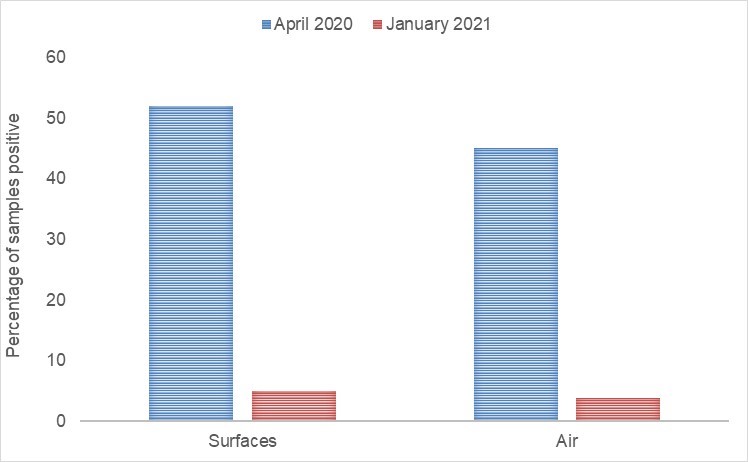
Way back during the first wave of COVID-19 (April 2020), we got our swabs out and sampled surfaces and air across a range of settings in the hospital. This cross-sectional study identified SARS-CoV-2 RNA on a rather startling 52% of surfaces and 45% of air samples collected from across the hospital and some public areas. During the second wave (January 2021), we undertook a similar exercise, and identified a vastly different level of contamination with SARS-CoV-2 RNA: 5% of surfaces and 4% of air samples. What had changed between April 2020 and January 2021 to explain this difference? A whole host of things, and we don’t know for sure – but I suspect that improved testing availability and the introduction of masks for staff and patients were the most important factors.
Right at the start of the pandemic, when there was chaos and disorder all around (bordering on panic at times!) we somehow managed to find a bit of time to get out into a range of settings in our hospitals to sample surfaces and air. At this time, very little was known about the transmission dynamics of SARS-CoV-2, the role of surface contamination in spread, and the relative importance of surface and air contamination. We certainly didn’t have to go far to find contamination with SARS-CoV-2 RNA! We identified surface and air contamination in every single one of the areas that we sampled, including the adult emergency department, an acute admissions ward, two COVID-19 cohort wards, three theatres during tracheostomy procedures, an intensive care unit, a ward with a 6-bedded bay converted into a negative pressure area for management of continuous positive airway pressure (CPAP) on patients with COVID-19, and the entrance and public area of a main hospital building. In fact, over half of the surface samples and almost half of the air samples were positive for SARS-CoV-2 RNA. Importantly, though, no live virus was cultured (and we tried with every single sample). This made the findings close to uninterpretable in terms of influencing policy and IPC approach. Was the SARS-CoV-2 RNA simply a marker of shedding or did it represent a live transmission risk? How much of our failure to detect live virus was influenced by laboratory factors?
When the second wave came in the winter of 2020/21, we managed to get out to sample surfaces and air in comparable areas. We were curious to see whether the levels of surface and air contamination would be similar between the two waves, whether the emergence of other variants would affect the profile of surface or air contamination, and whether we could discern any role for ventilation in mitigating air (and indirectly surface) SARS-CoV-2 contamination.
The comparison overall was pretty stark (see Figure 1), with around 10-fold less surface and air contamination identified in the second vs. the first wave. As in the original study, no live virus was detected from any of the samples. So what could have explained the reduction? Since both studies were cross-sectional and not longitudinal, we don’t know how much the level of contamination varied from day to day. It could have been if we’d sampled on different days, we would have seen a very different picture. However, it seems much more likely that the difference in infection prevention and control measures implemented between April 2020 and January 2021 explain the difference. These difference include changes to patient management (including more testing and mask wearing), staff (masking, 2x weekly lateral flow testing, and early roll out of vaccination), visitor restrictions, and attempts to improve ventilation. It’s pure speculation to say which of these is most important – but I’m going to do it anyway! I suspect testing and masking for staff and patients were the most important factors.
Figure 1: Percentage of surface and air samples positive for SARS-CoV-2 RNA in April 2020 vs. January 2021. 218 surfaces samples were collected in April 2020 and 270 in January 2021; 27 air samples were collected in both April 2020 and January 2021.
A key theme of the pandemic has been the role of ventilation in preventing the spread of respiratory viruses. In order to evaluate whether ward-level ventilation influences air (and indirectly surface) contamination, we did rounds of sampling in two wards that had no mechanical ventilation whatsoever, but (through necessity) were being used as COVID-19 cohort wards. You might expect the levels of air and surface contamination to be considerably higher in these wards. However, this was not the case, and we didn’t identify a significantly higher level of contamination in these wards without mechanical ventilation. How do we interpret this? Well, whilst the other wards did have some degree of mechanical ventilation, we didn’t measure airflows and it’s fair to say the mechanical ventilation isn’t the best. Also, natural ventilation isn’t the same as no ventilation, and again since we didn’t measure airflows, it’s difficult to make conclusions from this part of the study. That said, on the face of it, it suggests that COVID-19 from the patients in the ward didn’t fill the air in the space in a way that something like cigarette smoke would.
Finally, we were able to do some genotyping of SARS-CoV-2 from patients in the vicinity of where the samples were taken, and of the SARS-CoV-2 from the environment, and concluded that the majority was the fairly recently emerged Alpha variant (remember that?!). We also performed a laboratory evaluation, and found that we could recover viable SARS-CoV-2 from dried surfaces for at least 72 hours after inoculation, supporting at least a theoretical role of dry surfaces in the spread of SARS-CoV-2, as we see with other respiratory viruses (like influenza).
Limitations of the study include:
- Design was cross-sectional rather than longitudinal – we can’t comment on day-to-day variation in the levels of surface and air contamination, and have to assume that our findings are representative of any given day.
- Samples were not collected from patients, air, and surfaces at the same time, meaning that we cannot link contamination levels to individual patients.
- We can’t rule out that our failure to identify live virus was due to methodological issues (although the CT values from the PCRs support that the virus was likely to be non-viable).
- We didn’t sample exactly the same areas, and there was some small variation in the exact spots that we took samples from. This was driven mainly by changes in use of clinical areas, and we tried to match like-for-like where possible.
- The work was done pre-Omicron – so perhaps we should get our swabs out again!
We identified a substantially lower level of surface and air contamination of clinical and public areas of a London hospital in the first vs. second waves of the COVID-19 pandemic. The reasons for this difference are not clear, but are probably explained by enhanced infection prevention measures. Whilst it may be true that these measures reduced surface and air contamination, they did not eliminate transmission risk, because hospital-onset COVID-19 infection continued to occur during the second wave. Finally, our findings suggest that ward-level ventilation is not the key determinant for high levels of air and surface contamination with SARS-CoV-2.
Discover more from Reflections on Infection Prevention and Control
Subscribe to get the latest posts sent to your email.