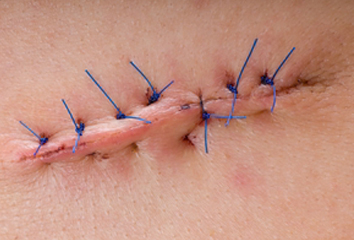
I love and hate SSI prevention in equal measure. On the one hand, we have good evidence and strong guidelines around SSI prevention – but on the other hand, implementing these interventions to prevent SSI can be enormously tricky in practice. One key factor in preventing SSI is creating a well-closed wound in theatre. Surprisingly, there’s precious little data on how to measure what a well-closed surgical wound looks like. This qualitative study presents some potential indicators to measure good surgical wound closure in order to prevent SSI.
wound infection
Can a wound dressing colour change indicate infection?
For many years, diagnostic labs have used colour change as a marker for the growth of specific microbes. Think of all those chromogeneic agar plates that your lab goes through each day. And there are all sorts of broths that change colour in response to specific chemical changes caused by microbial growth. One of the first projects I was ever involved with was environmental sampling for MRSA at Lewisham hospital, where we used a selective broth that turned bright yellow when MRSA was present. So in a way, it is surprising that this approach has not been adopted as a marker to indicate wound infection.
Scientists at Bath Uni have developed a neat novel wound dressing that fluoresces when the early signs of an infection are present. The concept is simple: a gel containing tiny sacs of dye are included in a hydrogel dressing; if cytotoxins that are indicative of bacterial infection are present, they lyse the sacs and release the dye, which fluoresces under UV light.
Chronic wound? No problem – a splash of oxygen peroxide should do the trick
I was involved in a recently published RCT of a novel wound care system (‘BioxyQuell’), which trailed the application of an aqueous oxygen peroxide (AOP, aka aqueous ozone) lavage on venous leg ulcers. The study isn’t perfect, but the results are encouraging and should serve as a basis for further evaluations.
The RCT was performed in the community setting. Sixty-one patients were enrolled if they had chronic venous leg ulcers, and randomised to either 6 AOP treatments or sham placebo treatments with sterile water over 2 weeks. AOP and placebo arms were both treated with standard-of-care compression bandaging between treatments and for the duration of the study. Patients who completed the initial 8 week RCT were enrolled into a follow-up study evaluating wound healing at 12 weeks, 6 and 12 months. The key results are:
- No significant difference in wound healing at 8 weeks (the primary outcome). But, something surprising happened during the follow-up study: every single one of the AOP treated patients had healed by 12 months vs. 50% in the placebo arm (Figure 1). Could it really be that a 2 week AOP treatment period has such a remarkable impact on long-term wound healing? It’s just about plausible if there was some pretty fundamental change to the wound bed. But I’d need to see some more data to be convinced!
- Almost 50% of patients in the placebo arm were healed by 8 weeks. The inclusion criteria required a chronic leg ulcer more than 3 months old, so why did 50% of placebo patients heal? I suspect the answer lies in good compression bandaging, which was mandated during the trial. There’s a temptation to apply the bandages too loosely to be effective to improve patient comfort.
- AOP patients were significantly less likely to be withdrawn from the study due to wound infection during the first 4 weeks of the RCT (Figure 2). This stark difference makes sense: AOP is a biocide with the capacity to dismantle biofilms, which are involved in wound infection.
- Pain and bacterial contamination were significantly lower in the AOP arm during the treatment phase. Wound size was also significantly reduced in the AOP arm when accounting for patients with deteriorating infected wounds.
Figure 1: Wound healing in the AOP and placebo arm during the RCT (up to 8 weeks) and follow-up study (up to 12 months).
Figure 2: Patient withdrawals due to wound infection during the first 8 weeks of the RCT.
As ever, the study raises as many if not more questions than it answers:
- What is the optimal treatment regimen for AOP? Is two weeks enough? Is three treatments per week about right (and feasible)? Put another way, how would this study have looked if the AOP treatment was performed for 4 weeks instead of 2? The fact that improvements in wound size reduction, pain, bacterial contamination and withdrawal due to wound infection trailed off after the 2 week treatment period indicates that a longer treatment period should be explored.
- What is the best application of AOP in healthcare? Acute care? Primary care? Trauma? Diabetic foot?
- Is ‘wound healing’ the right outcome measure? Perhaps prevention of withdrawals due to wound infection is the most important finding of this study. And don’t underestimate the value of the reduced pain in the AOP arm – the ulcers are very painful and make sufferers pretty miserable!
This was a fair sized randomised, sham placebo controlled, double blinded study performed with tight inclusion and exclusion criteria in the community setting. This sort of RCT is surprisingly rare in the wound care literature. However, it was terminated early (due to lack of significant difference in wound healing and the fact that it took years to recruit a decent number of patients) and the primary outcome (wound healing at 8 weeks) was non-significant. Nonetheless, wound size, pain and bacterial contamination were reduced by AOP treatment, the long-term reductions in wound healing are tantalizing, and reduced withdrawals due to wound infection are tangible.
Post script: in memoriam
The statistician who formulated and executed the statistical analysis for this study, Carla White, sadly died (too young) during the latter stages of getting this article published. I am sorry not to have the opportunity to work with her again.