The old dogma to “always complete your antibiotic course” has been challenged recently, see BMJ and previous blogs. Is it safe to tell patients to stop whenever they feel better? Purely by coincidence this paper appeared, and was discussed in our PhD’s Journal Club. The paper’s title was Individualizing duration of antibiotic therapy in community-acquired pneumonia (CAP), and the students were surprised by the final result, reports Valentijn Schweitzer.
The study: A multicenter non-inferiority randomized controlled trial in which CAP patients that reached clinical stability within 5 days were randomized to standard care or a group in which antibiotics were stopped if patients were clinically stable (and if clinically stable for at least 48 hours). A composite endpoint for failure (which included death) within 30 days was the primary outcome.
As part of protocol they also defined stopping criteria for safety, which were (1) inferiority of the individualized treatment arm or (2) proven harm for patients in both arms. Think a few seconds of the latter: how would you proceed in treating patients?
After randomizing 260 patients (125 with individualized treatment) the primary outcome (failure) was reached in 14 (11.4%) with individualized treatment and in 10 (7.4%) controls (p=0.20). On average, they had received antibiotics for 6 and 8 days (p<0.001). Yet, the Data Safety Monitoring Board (DSMB) decided to terminate the trial (that was planned for 950 patients) based on observed 30-day mortality (4 in individualized arm and I in control). Yet, according to the blinded adjudication committee none of these casualties were related to CAP. Unclear how that knowledge contributed to the decision. Fun fact: many similar interventions are implemented as antibiotic stewardship without even assessing clinical outcomes.
In their discussion the PhD candidates were immediately lost in debating inferiority and non-inferiority. For this study: Should the stopping rule for an interim analysis in a non-inferiority trial on safety be based on inferiority or non-inferiority? Inferiority within the non-inferiority margin – at interim (as in this study) – might still result in non-inferiority with a full sample size, while reaching non-inferiority – at interim – is almost impossible (that’s why these studies need so many patients). The students concluded that a conditional power analysis on futility of non-inferiority might be the best option. (Clever students; they mean that you predict – at interim – the likelihood that non-inferiority still can be reached with the full sample size).
Although the authors try to save their work by concluding that an “early stop might not be safe”, the students learned that stopping criteria for a DSMB should be better defined or your trial may not justify any conclusion.
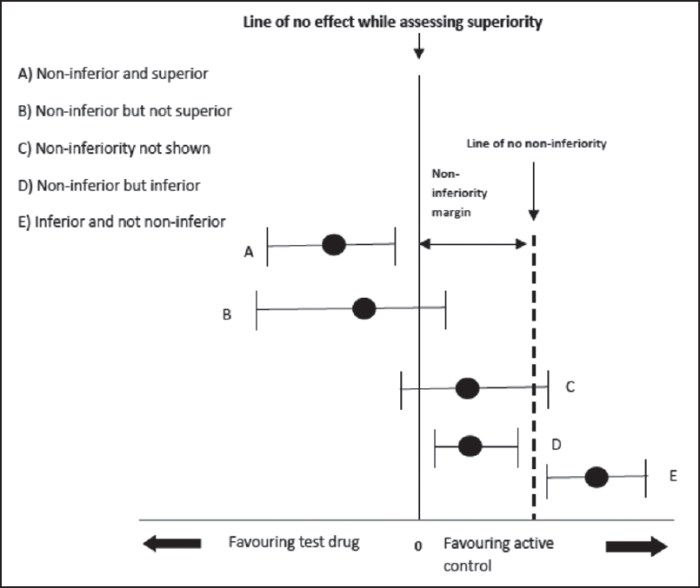
They needed this picture (that says more than a thousand words) of possible outcomes of a non-inferiority trial to end their discussion.

Valentijn Schweitzer does a PhD on effectiveness of antibiotic stewardship in patients with CAP
Discover more from Reflections on Infection Prevention and Control
Subscribe to get the latest posts sent to your email.